Small molecule factors found to influence age-related bone deterioration
Posted: 18 September 2018 | Drug Target Review | No comments yet
Scientists in the U.S. have reported findings about the mechanisms that contribute to the experience of weaker bones with age.
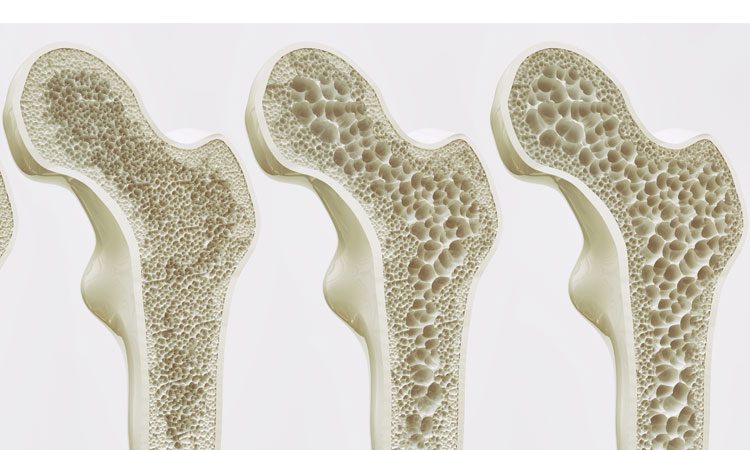

The published report explains that expression of small molecule microRNA-141-3p, which silences other molecules, increases dramatically while a key signalling molecule, stromal-cell derived factor or SDF-1, that helps stem cells make healthy bone decreases.
The researchers found evidence in both mouse and human mesenchymal stem cells that this unhealthy shift happens, and that correcting it can result in healthier bone formation.
Commenting on the significance of small molecule levels, Dr Sadanand Fulzele, bone biologist in the Department of Orthopaedic Surgery at the Medical College of Georgia at Augusta University, said: “If you are 20 years old and making great bone, you would still have microRNA-141-3p in your mesenchymal stem cells. But when you are 81 and making weaker bone, you have a lot more of it.”
Restoring a more youthful balance could constitute a novel strategy for reducing age-associated diseases such as osteoporosis and the impaired ability to heal bone breaks, explained Fulzele, a corresponding author on the study.
“You want it sort of in that sweet spot,” says Dr William D. Hill, a longtime stem cell researcher at MCG now on the faculty at the Medical University of South Carolina. “What we are trying to do is dial it back down from where it’s being overexpressed due to factors like ageing and oxidative stress and suppression of oestrogen, and bring it back into a range that would effectively allow more normal bone formation,” says Hill, also a corresponding author.
The International Osteoporosis Foundation claims that approximately 30% of postmenopausal women in the U.S. and Europe have osteoporosis, and at least 40% of these women and 15-30% of men will sustain one or more fractures in their lifetime – one fracture placing them at increased risk of others.
Mesenchymal stem cells can differentiate into the major components of our skeleton: bone-forming osteoblasts; actual bone cells or osteocytes, made by osteoblasts; cartilage cells called chondrocytes; as well as fat cells, or adipocytes.
SDF-1 is a key signalling molecule that helps regulate the differentiation of stem cells into these cells, the MCG research team has shown. SDF-1 has a myriad of other roles as well, including helping mesenchymal stem cells get to the right spot during bone formation and bone repair and protecting cells from the ravages of oxidative stress.
Their animal studies had indicated that oxidative stress in mesenchymal stem cells decreases SDF-1 and that the signalling molecule could protect those cells from death by oxidative stress.
Piecing together their findings the scientists found that higher oxidative stress elevates microRNA-141-3p expression, which in turn decreases SDF-1 levels.
In both mice and human mesenchymal stem cells, they found levels of microRNA-141-3p were low in young cells but tripled or more in older cells. They found the opposite to be the case for SDF-1 levels.
When they injected a microRNA-141 mimic inside the stem cells, it essentially created a model of ageing and SDF-1 levels again went down. Consequences of that included another shift that normally occurs with age as we make more bone-eating osteoclasts than bone-forming osteoblasts. The shift also resulted in mesenchymal stem cells instead making more fat, which they tend to do with age because it’s easier.
In a bid to test their hypothesis from all angles, the scientists also added microRNA-141-3p to cells and watched bone function get worse, then used the inhibitor again and saw improvement.
Clinical-grade drugs, like the research drug they used to inhibit microRNA-141-3p and that might also target other members of the micro-RNA 141 family, could one day be an effective way to help mesenchymal stem cells remain focused on making bone in the face of age and other conditions, said Fulzele.
In their cell studies: “It normalises bone function. We think clinical-grade inhibitor may help us do the same in people,” he said.
They are now looking to move into animal models and assess a wide array of other factors, such as how fat production is influenced and whether they can improve fracture healing in a model of ageing and/or prevent or at least reduce osteoporosis. Fulzele also wants to find out if higher physical activity levels, which tend to diminish with age, can also help restore a healthier balance of microRNA-141-3p and SDF-1.
Even normal, healthy ageing results in increased levels of oxidative stress, which includes things like reactive oxygen species that are byproducts of oxygen use.
The human stem cells the scientists isolated and analysed came from 18-40 year olds and 60-85 year olds who had orthopaedic surgery. The study was reported in the Journal of Gerontology: Biological Sciences.
Related topics
Disease Research, microRNA, Small molecule
Related conditions
Osteoporosis
Related organisations
Medical College of Georgia at Augusta University, Medical University of South Carolina (MUSC)
Related people
Dr Sadanand Fulzele, Dr William D. Hill