Potential new therapeutic target for Alzheimer’s discovered
Posted: 18 June 2019 | Drug Target Review | No comments yet
New study finds a promising antagonist that blocks specific interaction between the protein apoE and amyloid precursor protein.
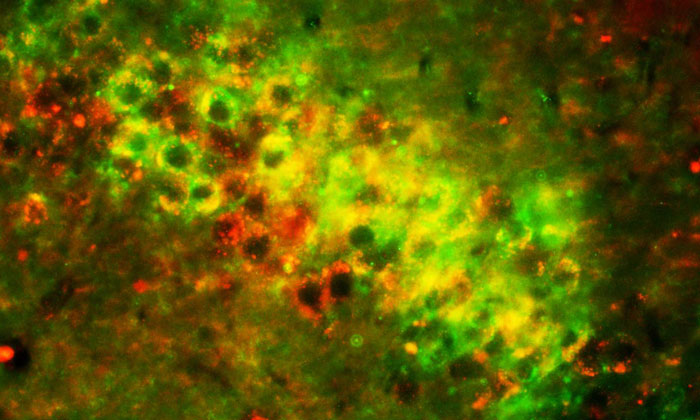

This microscopic image shows the merging of APP and apoE in brain cells. This co-localization suggests an age-associated increase in apoE-N-terminal APP interaction and thus higher production of the toxic amyloid-β (Aβ) protein characteristic of Alzheimer's disease (© University of South Florida).
A new study has shown that a novel apoE antagonist blocks apoE interaction with N-terminal amyloid precursor protein (APP). Moreover, this peptide antagonist (6KApoEp) was shown to reduce Alzheimer’s-associated beta amyloid (β-amyloid) accumulation and tau pathologies in the brain. It also improved learning and memory in mice genetically engineered to mimic symptoms of Alzheimer’s disease.
Many failed anti-amyloid therapies for Alzheimer’s disease have been directed against various forms of the protein β-amyloid, which ultimately forms clumps of sticky plaques in the brain. The presence of these amyloid plaques is one of the major hallmarks of Alzheimer’s disease.
The research, which was led by the University of South Florida Health (USF Health) Morsani College of Medicine, suggests that disrupting apoE physical interaction with N-terminal APP may be a new disease-modifying therapeutic strategy for this most common type of dementia.
“For the first time, we have direct evidence that the N-terminal section of apoE itself acts as an essential molecule (ligand) to promote the binding of apoeE to the N-terminal region of APP outside the nerve cell,” said the study’s lead author Darrell Sawmiller, PhD, an assistant professor in the USF Health Department of Psychiatry and Behavioral Neurosciences. “This receptor-mediated mechanism plays a role in the development of Alzheimer’s disease. Overstimulation of APP by apoE may be an earlier, upstream event that signals other neurodegenerative processes contributing to the amyloid cascade.”
“Initially we wanted to better understand how apoE pathologically interacts with APP, which leads to the formation of β-amyloid plaques and neuronal loss,” added study senior author Jun Tan, PhD, MD, a professor in the USF Health Department of Psychiatry & Behavioral Neurosciences. “Our work further discovered an apoE derivative that can modulate structural and functional neuropathology in Alzheimer’s disease mouse models.”
The preclinical study was published online in Biological Psychiatry.
Related topics
Disease Research, Drug Targets, Protein, Research & Development
Related conditions
Alzheimer’s disease
Related organisations
University of South Florida Health (USF Health) Morsani College of Medicine
Related people
Darrell Sawmiller PhD, Jun Tan PhD MD