Potential ovarian cancer treatment proves effective in pre-clinical studies
Posted: 14 June 2021 | Victoria Rees (Drug Target Review) | No comments yet
A new radiopharmaceutical compound has shown success at treating ovarian cancer in mice and in vitro studies.

Pre-clinical trials of a new radiopharmaceutical therapy to treat ovarian cancer have produced successful results, according to a new study. Conducted at Michigan State University, US, the researchers from the institute and Niowave Inc, found the agent dramatically limited tumour growth and decreased tumour mass in models.
Designed specifically for ovarian cancers that are resistant to traditional therapies, the new radiopharmaceutical can be produced in 25 minutes at low cost, which leads to better efficiency compared with alternative methods, the team say.
In the study, the researchers utilised a new generator system to develop the targeted alpha therapy Pb-214-TCMC-trastuzumab to treat HER2-postive ovarian cancer. Ovarian cancer cells and mice bearing ovarian cancer tumours were split into three groups: those treated with Pb-214-TCMC-trastuzumab, those treated with Pb-214-TCMC-IgG and an untreated control group. All groups were imaged over time to determine the effectiveness of the treatment.
Compared to the Pb-214-TCMC-IgG and control groups, the team found that the tumour signal for mice and cells treated with Pb-214-TCMC-trastuzumab decreased dramatically over the course of the study, signalling the efficacy of the therapy. There were no adverse side effects from the treatment as determined by weight loss of all animals surviving.
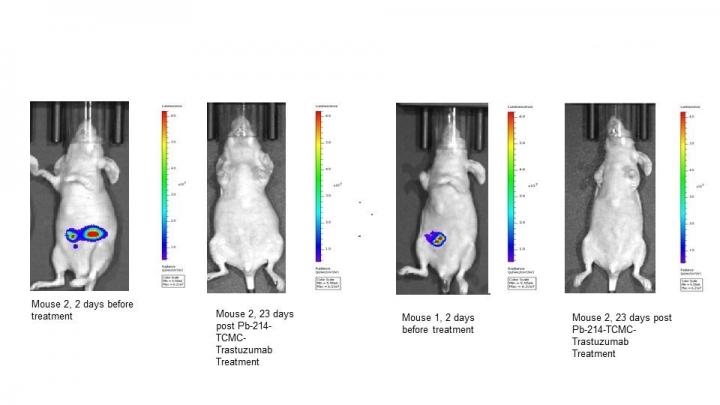

Bioluminescence images overlaid with pictures of representative mice implanted intraperitoneally with luciferase-positive ovarian cancer cells (SKOV3) before and after treatment with Pb-214-TCMC-trastuzumab [credit: Abdullah Metebi, PhD Student at the Institute for Quantitative Health Science and Engineering, Michigan State University].
“The short 27-minute half-life of Pb-214 is ideal for fractioned alpha particle therapeutic applications,” said Mike Zamiara, study author and president of Niowave. “The generator system can provide Pb-214 every hour, potentially providing a new source of alpha particle therapy to patients at lower cost. In the future, the generator system will be available for many therapeutic products in a turn-key system under development, providing reliable doses for improved patient care.”
The research from this study was presented at the Society of Nuclear Medicine and Molecular Imaging 2021 Annual Meeting.
Related topics
Drug Development, Drug Discovery, Oncology, Target Validation, Therapeutics, Translational Science
Related conditions
Ovarian cancer
Related organisations
Michigan State University, Niowave Inc
Related people
Mike Zamiara