Tweaked bacteria genes activate heart cells in mice
Posted: 7 February 2022 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have shown that engineered bacterial genes coding for sodium ion channels could lead to novel gene therapies for electrical heart diseases.

Biomedical engineers at Duke University, US have demonstrated that a new gene therapy can help heart muscle cells electrically activate in live mice. According to the team, the approach features engineered bacterial genes that code for sodium ion channels and could lead to therapies to treat a wide variety of electrical heart diseases and disorders.
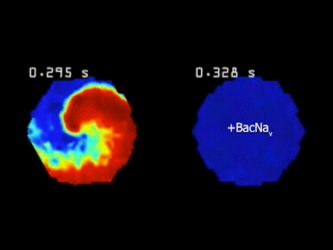

Cardiac arrhythmias occur when heart muscle cells do not uniformly transmit electrical signals to pump blood in a cohesive, orderly fashion. The left shows arrhythmic cells in tachycardia chaos, whereas the right shows cells treated with the new gene therapy behaving normally, as they are much more difficult to push out of their regular heart beat activity [credit: Tianyu Wu, Duke University].
“We were able to improve how well heart muscle cells can initiate and spread electrical activity, which is hard to accomplish with drugs or other tools,” said Professor Nenad Bursac, one of the authors of the paper published in Nature Communications. “The method we used to deliver genes in heart muscle cells of mice has been previously shown to persist for a long time, which means it could effectively help hearts that struggle to beat as regularly as they should.”
Sodium-ion channels are proteins in the outer membranes of electrically excitable cells, such as heart or brain cells, that transmit electrical charges into the cell. In the heart, these channels tell muscle cells when to contract and pass the instruction along so that the organ pumps blood as a cohesive unit. Damaged heart cells, however, whether from disease or trauma, often lose all or part of their ability to transmit these signals and join the effort.
One approach to restore this functionality is gene therapy. By delivering the genes responsible for creating sodium channel proteins, the technique can produce more ion channels in the diseased cells to help boost their activity.
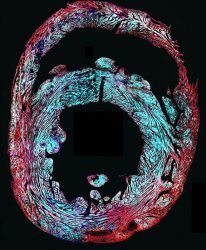

This cross-section of a mouse heart (red) shows how well the gene therapy delivered sodium ion channel genes (cyan) to the target heart cells after researchers injected a virus with the genes into the mouse veins [credit: Tianyu Wu, Duke University].
In mammals, sodium channel genes are too large to fit within the viruses currently used in modern gene therapies in humans. To skirt this issue, the team utilised smaller genes that code for similar sodium ion channels in bacteria. While these bacterial genes are different than their human counterparts, evolution has conserved many similarities in the channel design since multi-cellular organisms diverged from bacteria hundreds of millions of years ago.
These genes were mutated so that the channels they encode could become active in human cells. The content of the genes was then optimised and combined with a “promoter” that exclusively restricts channel production to heart muscle cells. The researchers tested their approach by delivering a virus loaded with the bacterial gene into veins of a mouse to spread throughout the body.
“We worked to find where the sodium ion channels were actually formed and as we hoped, we found that they only went into the working muscle cells of the heart within the atria and ventricles,” said doctoral student Tianyu Wu, one of the researchers on the study. “We also found that they did not end up in the heart cells that originate the heartbeat, which we also wanted to avoid.”
The researchers highlight that this gene therapy approach only delivers extra genes within a cell; it does not attempt to cut out, replace or rewrite the existing DNA in any way. Scientists believe these types of delivered genes make proteins while floating freely within the cell, making use of the existing biochemical machinery. Previous research with this viral gene delivery approach suggests the transplanted genes should remain active for many years.
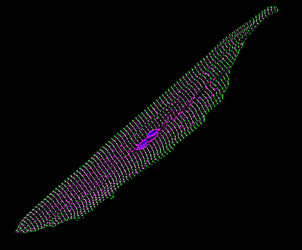

This detailed image of a single mouse heart muscle cell shows its cell membrane expressing the new sodium ion channel genes (magenta) after researchers delivered the therapy through an injection into the mouse veins [credit: Tianyu Wu, Duke University].
As a proof-of-concept, tests on cells in a laboratory setting suggest that the treatment improves electrical excitability enough to prevent human abnormalities like arrhythmias. Within live mice, the results demonstrate that the sodium ion channels are active in the hearts, showing trends toward improved excitability. However, the team say further tests are needed to measure how much of an improvement is made on the whole-heart level and whether it is enough to rescue electrical function in damaged or diseased heart tissue to be used as a viable treatment.
Moving forward, the researchers have already identified different bacterial sodium channel genes that work better in preliminary benchtop studies.
“I think this work is really exciting,” Bursac said. “We have been harnessing what nature made billions of years ago to help humans with modern-day disease.”
Related topics
Drug Delivery, Gene Therapy, Microbiology, Translational Science
Related organisations
Duke University
Related people
Professor Nenad Bursac, Tianyu Wu