New imaging technique may predict the of spread breast cancer
Posted: 16 August 2017 | Dr Zara Kassam (Drug Target Review) | No comments yet
Researchers have developed a radioactive ‘tracer’ molecule to detect myeloid-derived suppressor cells’ accumulating in the lung in preparation for the arrival of breast cancer cells…
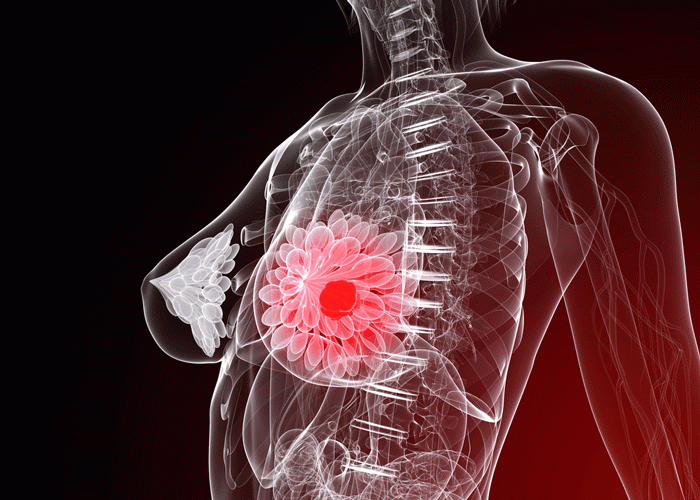
Researchers have developed a radioactive tracer molecule to detect myeloid-derived suppressor cells’ (MDSCs) accumulating in the lung in preparation for the arrival of breast cancer cells and the formation of metastases.
By using this new non-invasive imaging method, they can detect changes in the lungs that signal breast cancer may soon spread there – before any metastases are visible. If validated in humans, this approach could enable patients to be offered more intensive therapy earlier, to potentially prevent breast cancer spread. The study has been published in Theranostics,
Previous research has shown that the gathering of a special type of immune cell MDSCs – in locations such as the lung – prepares the ground for breast cancer metastasis, by suppressing the local immune system and promoting the formation of angiogenesis.
The research teams – led by Dr Michel Eisenblätter and Dr Fabian Flores-Borja at King’s College London – initially hypothesised that, by making the protein S100A8/A9 visible, it could be possible to identify when the lungs were being prepared for colonisation by breast cancer cells.
To test this theory, they developed a tracer antibody able to attach itself to S100A8/A9, that can be detected using a special scan called SPECT (single photon emission computed tomography) – a 3D gamma-ray scanner. The tracer releases radiation which can be distinguished by the SPECT scanner, highlighting areas where S100A8/A9 molecules, and MDSCs, are accumulating.
Previous research has shown that the gathering of a special type of immune cell called ‘myeloid-derived suppressor cells’ (MDSCs) – in locations such as the lung – prepares the ground for breast cancer metastasis, by suppressing the local immune system and promoting angiogenesis.
The researchers tested the tracer with triple negative breast cancer cells that had been implanted in mice, and were able to show – using a miniature SPECT scanner – that the ‘tracer’ lit up the lungs of mice well before any tumour cells were visible in the lung. Furthermore, the tracer could distinguish between highly aggressive breast cancer with a high tendency to spread to the lung and tumours without metastatic potential.
Currently, to assess the risk of a patient’s breast cancer spreading, doctors use tools based upon the features of the primary tumour, such as tumour size and whether it has already invaded nearby lymph nodes. With this early-stage study demonstrating the technique to be effective in mice, further studies are now required to develop a more effective ‘tracer’ molecule – better suited for use in humans – to be tested in clinical trials in future.
Dr Flores-Borja said: “By combining cell biology and imaging techniques, we have established a method to predict, at an early time-point during tumour development, whether tumour invasion will occur. “We envision this technique being used to help select patients for either further surveillance or intensified therapy, as well as aiding cancer research. The development of a test that is able to identify an increased risk of metastasis soon after a patient is diagnosed with breast cancer, would be very useful in helping choose the best treatment for patients.”
Baroness Delyth Morgan, Chief Executive at Breast Cancer Now, which helped to fund the study, said: “While more research is needed before this could be tested in patients, the prospect of a hospital scan which could predict whether breast cancer will spread to the lungs is incredibly exciting.
“This is a promising step towards being able to use 3D imaging to help offer more personalised therapy. Ultimately, anything that could provide patients and their doctors with a more accurate picture of whether their breast cancer may spread will help us tailor treatments to stop this from happening,” she added.
This work was also supported by the National Institute for Health Research Biomedical Research Centre at Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, KCL and UCL Comprehensive Cancer Imaging Centre, the German Research Foundation, Cancer Research UK, and the Engineering and Physical Sciences Research Council (EPSRC). The SPECT scanning equipment at KCL was funded by the Wellcome Trust.
Related topics
Antibodies, Imaging, Oncology
Related conditions
Breast cancer, Lung cancer
Related organisations
Cancer Research UK, German Research Foundation, King's College London, National Institute for Health Research Biomedical Research Centre, ngineering and Physical Sciences Research Council, University College London, Wellcome Trust
Related people
Baroness Delyth Morgan, Dr Fabian Flores-Borja, Dr Michel Eisenblätter