New avenues open for the treatment of thrombosis
Posted: 28 May 2019 | Drug Target Review | No comments yet
New study describes for the first time the structure of Ixolaris and its action on the inhibition of thrombus formation.
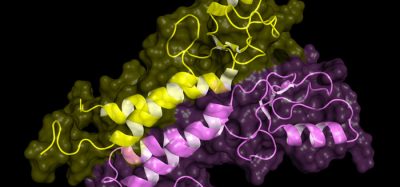
The new study by researchers from Brazil and the United States describes the Ixolaris structure, an important anticoagulant protein found in tick saliva, and its interaction with Factor Xa, a key enzyme in the process of blood clotting.
Conducted by Ana Paula Valente, Viviane de Paula, Robson Monteiro and Fabio C. L. Almeida, of the Federal University of Rio de Janeiro, in collaboration with Nikolaos Sgourakis, of the University of California and Ivo Francischetti from the National Institute of Health, this is the first description of the molecular mechanism of its action on the functioning of Factor Xa, an enzyme that plays a crucial role in the blood clotting.
Using nuclear magnetic resonance, the researchers were able to construct a 3D model of the structure formed by the association of these two proteins.
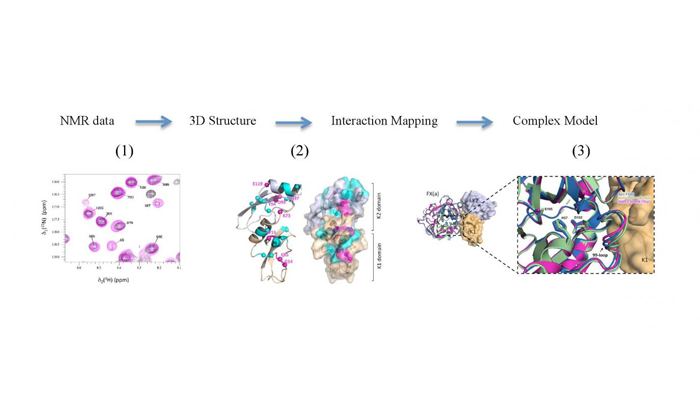
1) NMR data was used to the structure determination and 2) interaction site mapping and 3) all data was used to construct a model of the complex (credit: Ana Paula Valente).
The unprecedented result brings important insights into understanding the molecular mechanisms behind hemostasis, whose malfunction can lead to thrombosis. The expectation is that, from such a study, it will be possible to develop drugs capable of interrupting the formation of clots without interfering in the process of hemostasis as a whole.
The results may also contribute to the development of new treatments for thrombosis. “This 3D structure study opens up numerous possibilities for structure-based drug design targeting human coagulation factors that could be used as a therapeutic option to attenuate coagulation and inflammation associated with thrombosis,” said Viviane de Paula, first author of the work, professor at the Federal University of Rio de Janeiro and visiting scientist at UCSC.
The study ‘NMR structure determination of ixolaris and Factor X interaction 1 reveals a noncanonical mechanism of Kunitz inhibition’ has been accepted for publication in Blood and will be available online soon.
Related topics
Drug Development, Protein, Research & Development
Related conditions
thrombosis
Related organisations
California University, Federal University of Rio de Janeiro, National Institute of Health
Related people
Ana Paula Valente PhD, Viviane de Paula