How bacteria behind hospital infections block out antibiotics revealed
Posted: 3 September 2019 | Rachael Harper (Drug Target Review) | No comments yet
Scientists have shown that drug-resistant bacteria infections shut out antibiotics by closing tiny doors in their cell walls.
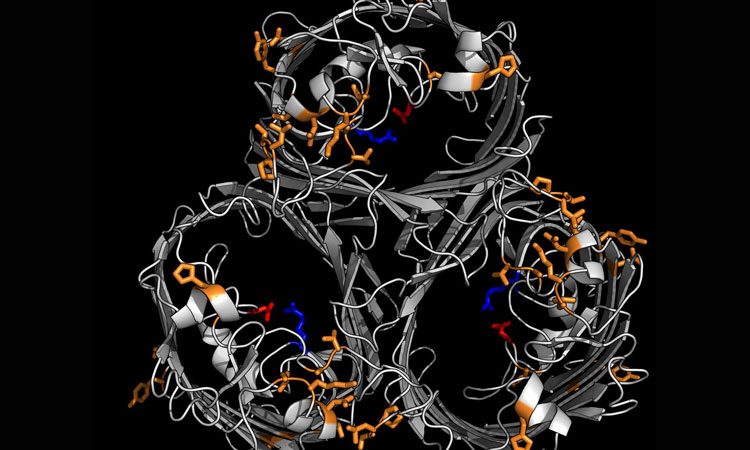

This is the structure of the antibiotic-resistant protein that closes the pore (door) to antibiotics trying to enter the bacterial cell (credit: Wong et al. 2019).
New findings by researchers at Imperial College London, UK, have shown that drug-resistant bacteria responsible for deadly hospital-acquired infections shut out antibiotics by closing tiny doors in their cell walls. According to the authors of the study, the discovery could allow researchers to design new drugs that allow antibiotics into bacterial cells.
The bacterium Klebsiella pneumoniae causes infections in the lungs, blood and wounds of those in hospitals. Like many bacteria, K. pneumoniae is becoming increasingly resistant to antibiotics, particularly Carbapenems. Carbapenems are used as antibiotics in hospitals when others have failed or are ineffective.
Now, the team from Imperial has discovered one mechanism by which K. pneumoniae is able to resist Carbapenems. Antibiotics usually enter the K. pneumoniae bacteria through pores. The team investigated the structure of the pores and showed that by shutting these doorways K. pneumoniae becomes resistant to multiple drugs, since antibiotics cannot enter and kill them.
The team compared the structures of K. pneumoniae bacteria that were resistant to Carbapenems to those that were not and found the resistant bacteria had modified or absent versions of a protein that creates pores in the cell wall. Resistant bacteria have much smaller pores, blocking the drug from entering.
This also means that the bacteria can take in fewer nutrients and tests in mice showed that the bacteria grew more slowly as a result.
However, the advantage in terms of avoiding antibiotics outweighed the negative impact of slower growth for the bacteria, allowing them to maintain a high level of infection.
“The modification the bacteria use to avoid antibiotics is difficult to get around,” said Professor Gad Frankel, from the Department of Life Sciences at Imperial, who led the team. “Any drugs to counteract this defence mechanism would likely also get blocked out by the closed doors.
However, we hope that it will be possible to design drugs that can pick the lock of the door and our data provides information to help scientists and pharmaceutical companies make these new agents a reality.”
As resistant bacteria are weaker, these results suggest that the pressure posed by the extensive use of Carbapenems in hospital settings is a major driver in the spread of these superbugs. The study provides a direct scientific basis for the implementation of restrictive prescribing policies that would minimise the use of broad-spectrum agents such as Carbapenems.
The results from the study was published in Nature Communications.
Related topics
Antibiotics, Cell Cultures, Drug Leads, Drug Targets, Research & Development
Related organisations
Imperial College London
Related people
Professor Gad Frankel