White blood cells as a novel early indicator of vaccine efficacy
Posted: 21 May 2020 | Hannah Balfour (Drug Target Review) | 1 comment
Researchers demonstrate that a subpopulation of circulating white blood cells can act as an early indicator of vaccine efficacy, with potential implications for COVID-19 drug development.
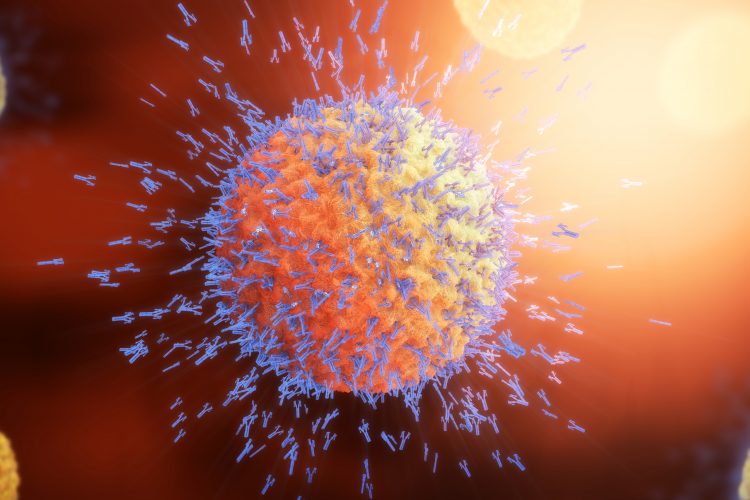

Researchers have established that a specific population of white blood cells found in circulation can be used as an early indicator of the efficacy of a vaccine. The team suggest their results could be significant to the development of a vaccine targeting COVID-19.
The team from Ludwig-Maximilians-Universität (LMU) in Germany were exploring the cellular immune responses to viral challenges and vaccines in order to aid in the development of novel preventatives and treatments for pathogenic viruses.
In their paper published in Clinical & Translational Immunology, the researchers demonstrated the quality of an immune response induced by vaccination can be ascertained after around seven days by measuring the frequency of a specific subclass of white blood cells in the circulation.
In their experiments the team used the vaccine for yellow fever, a live vaccine and one of the most effective on the market. Following vaccination with these attenuated, whole viral particles the virus replicates, inducing an immune response and the production of antibodies. The antibodies prevent the yellow fever virus from binding to and invading human cells.
“In our study, we monitored the development of the immune response in the blood of healthy subjects,” says study leader Dirk Baumjohann, who until recently was based in LMU’s Biomedical Center and is now a professor at Bonn University. “The individuals involved belong to a cohort of 250 people who had been vaccinated against yellow fever.”
The long-lived protection of the yellow fever vaccine is attributable to it triggering the production of very high levels of specific antibodies that can be detected decades after immunisation. Antibody formation and secretion requires Tfh cells, a subgroup of white blood cells primarily found in secondary lymphoid organs, including the lymph nodes and the spleen. “However, these organs are difficult to analyse in humans. But what are referred to as ‘Tfh-like cells’ are found in the circulation, and their frequencies correlate quite well with the relative concentrations of classical Tfh cells in secondary lymphoid organs,” explains Johanna Huber, a PhD student in Baumjohann’s group and lead author of the paper. “Therefore, analysis of circulating Tfh-like cells is relevant and can be performed directly in blood samples.” Despite this ease of access, the role of these circulating T cells in the immune response to the yellow-fever vaccine had not previously been examined in detail.
In the study, Baumjohann and his colleagues demonstrated that the properties and dynamics of Tfh-like cells enable the prediction of the efficacy of the immune response, even before detectable levels of neutralising antibodies (the typical measure of vaccine efficacy) appear in circulation. The formation of these highly potent neutralising antibodies in response to natural infection or vaccination can take up to several weeks. “In contrast, vaccine-induced T follicular helper cells can be quantified in the blood by about day seven after immunisation. In addition, we found that the frequency of a subpopulation of Tfh cells in the blood at two weeks after vaccination correlates with the quality of the antibody response that we detect a fortnight later,” Baumjohann said.
The authors suggested these findings may be of significance in developing a vaccine against COVID-19 and other viral diseases. Simon Rothenfusser, Department of Clinical Pharmacology, LMU Medical Center and Helmholtz Zentrum München added: “How SARS-Cov-2 triggers an immune response is essentially unknown at the moment. The vaccine against yellow fever is a paradigmatic example for the induction of live-long immunity against a specific virus. Our findings on the immune cell phenotypes in the yellow fever vaccine cohort will therefore provide a useful source of data for comparative studies on the immunogenicity of SARS-Cov-2 and other emerging classes of viral pathogens and vaccines for which it is not yet known if they induce a long-lasting protective immune response.”
Related topics
Antibodies, Biomarkers, Disease Research, Drug Development, Immunology, Vaccine
Related conditions
Coronavirus, Covid-19, viral infections, Yellow fever
Related organisations
Ludwig-Maximilians-Universität (LMU)
Related people
Dirk Baumjohann, Simon Rothenfusser






What happens to CBC particularly TLC &DLC.
What is the indication of a low TLC & low neutrophils after a week of covid vaccination.
Any association with increased CRP&ESR post vaccination.
What helps in diet&medication to improve immunity?