Over 200 genes implicated in glioblastoma tumour growth
Posted: 30 July 2020 | Victoria Rees (Drug Target Review) | No comments yet
A study has identified more than 200 genes that drive the progression of glioblastoma and which the researchers say present drug targets.
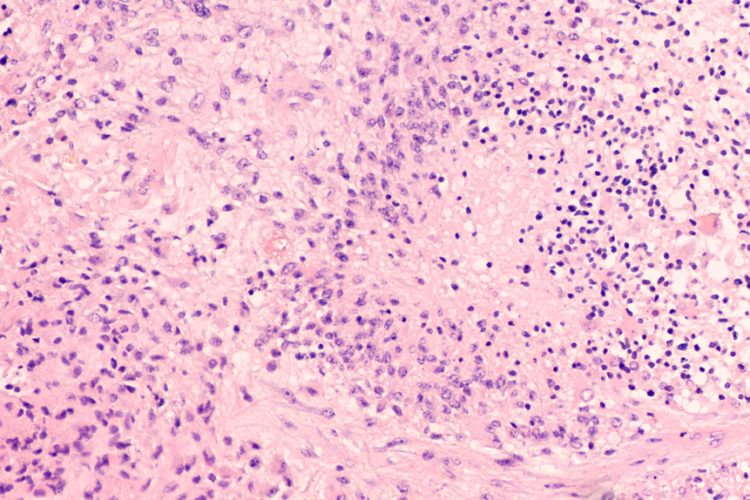

A study has identified more than 200 genes with novel and known roles in glioblastoma, which the researchers say offer promising new drug targets. The team, from the Wellcome Sanger Institute, Addenbrooke’s Hospital, both UK, and their collaborators, engineered a new mouse model to show for the first time how a mutation in the well-known cancer gene epidermal growth factor receptor (EGFR) initiates glioblastoma and works with a selection from more than 200 other genes to drive the cancer.
According to the researchers, the results present the first mouse model of its kind, which is available for the research community to advance new treatments for this lethal form of brain cancer. In the study, the model showed that EGFR alone can initiate brain tumours to grow in mice, resulting in tumours that were highly representative of human glioblastomas.
Dr Imran Noorani, a corresponding author previously from the Wellcome Sanger Institute, and now based at Addenbrooke’s Hospital and the University of Cambridge, said: “We have created a new mouse model for studying the lethal human brain cancer, glioblastoma. For the first time, we showed that the familiar cancer gene EGFR is capable of initiating glioblastoma and we identified new driver genes, whose potential for therapeutic targeting deserves further exploration.”
To identify which genes help EGFR to drive the cancer, the team used the PiggyBac transposon technique – a small section of DNA inserted into different parts of the genome to introduce mutations. This revealed more than 200 known and novel mutations in tumour suppressor genes that were working with EGFR to drive brain tumour growth, many of which present new drug targets.
The team compared the results with human genome sequences from glioblastoma patients and uncovered many genetic mutations found in both humans and mice. Human genomic data contains many mutations implicated in glioblastoma, without clear indication of which specific mutations drive the cancer. With the new mouse model, the team were able to narrow down on which mutations drive glioblastoma, which they say will focus future drug development.
Professor Allan Bradley, previously Director of the Wellcome Sanger Institute, and now Chief Scientific Officer of Kymab and Professor in the Department of Medicine, University of Cambridge, said: “Glioblastoma patients urgently require new, targeted therapies. Unfortunately, glioblastoma tumours can become highly resistant to therapies that target specific molecules, as there are many other genetic drivers that can ‘take over’ progressing the cancer. This new mouse model provides the missing link to translate findings from new potential treatments tested on mice to clinical trials.”
The findings were published in Genome Biology.
Related topics
Disease Research, Drug Targets, Genetic Analysis, Oncology, Targets
Related conditions
Cancer, Glioblastoma
Related organisations
Addenbrooke's Hospital, University of Cambridge, Wellcome Sanger Institute
Related people
Dr Imran Noorani, Professor Allan Bradley