New gene therapy could treat dominant optic atrophy
Posted: 27 November 2020 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have found that an OPA1-targeted gene therapy can treat dominant optic atrophy in pre-clinical trials.

Scientists have developed a new gene therapy approach that they say offers promise for one day treating an eye disease that leads to a progressive loss of vision and affects thousands of people across the globe. The study was led by researchers at Trinity College Dublin, Ireland.
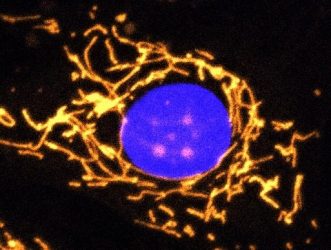

A fluorescent microscope image with mitochondria highlighted in gold. This healthy cell shows a highly elaborate and well-connected network of mitochondria [credit: Professor Jane Farrar and Dr Daniel Maloney, Trinity College Dublin].
According to the researchers, characterised by degeneration of the optic nerves, dominant optic atrophy (DOA) typically starts to cause symptoms in patients in their early adult years. There is currently no way to prevent or cure DOA.
The team say that the gene OPA1 provides instructions for making a protein that is found in cells and tissues throughout the body and which is pivotal for maintaining proper function in mitochondria, the powerhouse of cells. Without the protein made by OPA1, mitochondrial function is sub-optimal and the mitochondrial network which in healthy cells is well interconnected is highly disrupted.
For those living with DOA, it is mutations in OPA1 and the dysfunctional mitochondria that are responsible for the onset and progression of the disorder.
The scientists, led by Dr Daniel Maloney and Professor Jane Farrar, developed a new gene therapy, which successfully protected the visual function of mice who were treated with a chemical targeting the mitochondria and were consequently living with dysfunctional mitochondria.
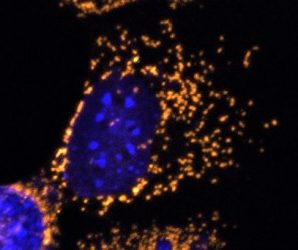

A fluorescent microscope image with mitochondria highlighted in gold. This cell completely lacks OPA1 protein and shows fragmented mitochondria [credit: Professor Jane Farrar and Dr Daniel Maloney, Trinity College Dublin].
The scientists also found that their gene therapy improved mitochondrial performance in human cells that contained mutations in the OPA1 gene, offering hope that it may be effective in people.
“We used a clever lab technique that allows scientists to provide a specific gene to cells that need it using specially engineered non-harmful viruses. This allowed us to directly alter the functioning of the mitochondria in the cells we treated, boosting their ability to produce energy which in turn helps protects them from cell damage. Excitingly, our results demonstrate that this OPA1-based gene therapy can potentially provide benefit for diseases like DOA, which are due to OPA1 mutations and also possibly for a wider array of diseases involving mitochondrial dysfunction,” said Maloney.
Farrar added: “OPA1 mutations are involved in DOA and so this OPA1-based therapeutic approach is relevant to DOA. However mitochondrial dysfunction is implicated in many neurological disorders that collectively affect millions of people worldwide. We think there is great potential for this type of therapeutic strategy targeting mitochondrial dysfunction to provide benefit and thereby make a major societal impact. Having worked together with patients over many years who live with visual and neurological disorders it would be a privilege to play a role in a treatment that may one day help many.”
The study was published in Frontiers in Neuroscience.
Related topics
Drug Targets, Gene Therapy, Genomics, In Vitro, In Vivo
Related conditions
Dominant optic atrophy (DOA)
Related organisations
Trinity College Dublin
Related people
Dr Daniel Maloney, Professor Jane Farrar