Scientists uncover structural biology details of SARS-CoV-2 infection
Posted: 26 February 2021 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have used computer simulations to model how the SARS-CoV-2 fusion peptide interacts with and penetrates the cell membrane.

New research in the lab of Emad Tajkhorshid at the University of Illinois, US, has addressed the molecular details of SARS-CoV-2 infection, which the researchers say could inform the design of drugs that block it.
According to the team, to infect cells, SARS-CoV-2 first attaches a molecule on the surface of cells, but then it has to fuse with human cells. A small region of the SARS-CoV-2 outer Spike (S) protein called the fusion peptide inserts itself into the human cell membrane to begin the fusion process. Scientists knew the location and approximate shape of the fusion peptide; however, they did not know exactly how it interacted with and penetrated into the human cell membrane and whether there would be changes in its shape when it stuck to the membrane. Without knowing the three-dimensional (3D) interactions between the SARS-CoV-2 fusion peptide and the cell membrane, it is not possible to design drugs that specifically disrupt that interaction.
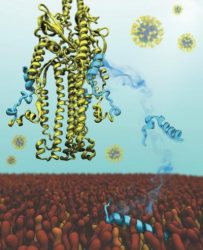

The SARS-CoV-2 virus inserts itself into the membrane of a host human cell using a small part of its S protein (yellow), called a fusion peptide. Computer simulations revealed the parts of peptide that are the most important for sticking to and inserting into the cell membrane. This illustration shows the fusion peptide (in blue) as it approaches, sticks to and inserts into the membrane (brown, orange and gold) [credit: Defne Gorgun].
Using computer simulations, the team merged what is known about the SARS-CoV-2 fusion peptide with the established 3D structures and behaviours of other coronavirus fusion peptides and simulated its interaction with a model human cell membrane. Their simulations reveal how the SARS-CoV-2 fusion peptide interacts with and penetrates the cell membrane.
“Our study shows which parts of the fusion peptide are important and how it sticks to and sits in the membrane,” said lead researcher Defne Gorgun, a graduate student.
As the model is theoretical, the researchers say the next step is to repeat their computer experiments in the lab with pieces of SARS-CoV-2 and cell membranes. However, having revealed parts of the fusion peptide that are likely to be critical to its function, those experiments will likely be completed faster and more efficiently. After that, the team say it will be possible to start testing drugs that disrupt the interaction and could help block SARS-CoV-2 from docking at cells.
Gorgun will presented her research on 25 February at the 65th Annual Meeting of the Biophysical Society.
Related topics
Drug Targets, Informatics, Protein, Proteomics, Structural Biology
Related conditions
Covid-19
Related organisations
University of Illinois
Related people
Defne Gorgun, Emad Tajkhorshid








