New 3D organoid model of pancreas developed from human stem cells
Posted: 29 April 2021 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have developed the first 3D organoid models of the pancreas that includes both the acinar and ductal structures.
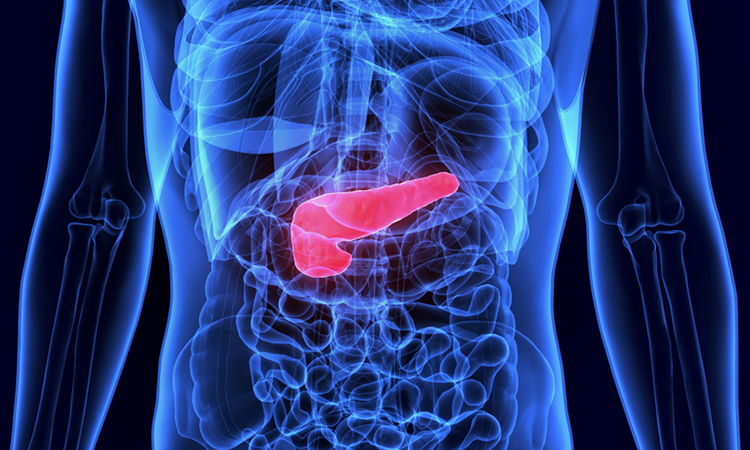

Researchers at the Cancer Research Institute at Beth Israel Deaconess Medical Center (BIDMC), US, have successfully created the first three-dimensional (3D) organoid models of the pancreas from human stem cells. Unlike previous platforms for the study of pancreatic cancer, this organoid model includes both the acinar and ductal structures that play a critical role in the majority of pancreatic cancers.
According to its developers, the novel research platform will shed new light on the origins and development of pancreatic cancer, as well as reveal potential means for discovering markers of early diagnosis and monitoring the disease.
“We thought, if we had a way to use human pancreatic cells to forward engineer cancer, we could begin to understand the earliest steps in the development of this disease,” said corresponding author Dr Senthil Muthuswamy. “This model could also serve as a platform to potentially discover biomarkers – measurable changes linked to disease – that we hope to use in the clinic to monitor cancer development.”
The pancreas is a hormone-secreting organ consisting of ducts and acinar cell structures. Researchers suspect that the most common kind of pancreatic cancer (pancreatic ductal adenocarcinoma, or PDAC) arises in the cells lining acinar and ductal structures. However, until now, scientists have not been able to successfully grow and maintain human acinar structures in the lab, challenging their ability to test the hypothesis in a model.
To coerce the stem cells down the path to becoming ductal and acinar cells, the researchers methodically tested various combinations of cell growth media used for different lengths of time. This study represents the first time researchers successfully generated human acinar cells in culture and maintained them long enough to be able to use them in experiments.
“It is like a cooking recipe – a complex recipe, with no precedent,” said Muthuswamy. “Only when you use all the right ingredients in the right proportions and do them in the correct sequence do you get the cells becoming the acinar cells.”
Next, the team used the two separate lineages of ductal and acinar organoids, engineered to include gene mutations known to be associated with pancreatic cancer. When the organoids were later implanted into mice, the different lineages behaved in distinct, predictable ways. For example, one mutation caused seven out of 10 of the mice transplanted with acinar-like organoids to develop cellular changes analogous to early pancreatic cancer in humans.
“Understanding the mechanisms that regulate these events will provide important insights into the events regulating the initiation of pancreatic cancer,” said Muthuswamy. “The idea is to see if we can identify biomarkers to benefit future patients – including by screening those at high risk for pancreatic cancer, so we can catch it before it is too late.”
The study was published in Cell Stem Cell.
Related topics
Disease Research, Oncology, Organoids, Stem Cells
Related conditions
Pancreatic cancer
Related organisations
Beth Israel Deaconess Medical Center (BIDMC)
Related people
Dr Senthil Muthuswamy