Treating cytokine storms in COVID-19 patients
Posted: 23 October 2020 | Dr Fiona McCann (CannBioRx Life Sciences), Dr H Michael Shepard (Enosi Life Sciences), Sir Marc Feldmann (Enosi Life Sciences) | No comments yet
Enosi Life Science researchers Sir Marc Feldmann, Dr H Michael Shepard and Dr Fiona McCann explain why anti-TNF therapies may be effective in treating COVID-19 associated cytokine storms and other inflammatory conditions.
Why anti-TNF therapy shows potential in treating COVID-19
Since the start of the COVID-19 pandemic, the novel coronavirus has been detected in nearly every country on earth. More than 35 million people worldwide have tested positive for the virus and over one million have died. Twenty percent of that number is in the US, which is disproportionately high.
The scale of the pandemic is reflected in the unprecedented efforts by industry and academia to develop a SARS-CoV-2 vaccine, with almost 200 vaccines in development by global players such as Johnson & Johnson, Sanofi, GlaxoSmithKline, Pfizer, Merck and some smaller companies such as Moderna. The world’s governments are committing eye-watering sums of money – in some cases billions – to vaccine development.
The delay of AstraZeneca’s clinical trial served as a painful reminder that vaccines do not come to fruition overnight and safety and efficacy must be proven. With a vaccine many months away and COVID-19 cases mounting, researchers are calling for every possible solution to be explored.
The discovery of high levels of inflammation and pro-inflammatory cytokines, such as tumour necrosis factor (TNF) and interleukin-6 (IL-6) in COVID-19 patients,1-3 has led researchers to evaluate blocking these mediators. Studies inhibiting IL-6 using receptor blockers such as tocilizumab or sarilumab4,5 have had limited success. However, anti-TNF therapeutics, which have a track record of successfully reducing inflammation in over 10 diseases and are the most widely used after corticosteroids, have not yet been properly evaluated.6,7
A closer look at inflammation in COVID-19
When a virus such as SARS-CoV-2 attacks the lungs, the body releases many mediators to fight the infection. There is evidence that interferons play an important role in this protective mechanism, as patients with abnormalities in this system or endogenous antibodies to interferon have poor outcomes.8,9 The high levels of pro-inflammatory TNF and IL-6 creates a “cytokine storm”, causing inflammation that may lead to severe respiratory distress or even death,10 partly because cytokine storms can also damage organs in addition to the lung.11
In COVID-19 cases, this hyperinflammatory response is characterised by higher-than-usual concentrations of serum TNF, IL-6 and IL-8, but relatively low IL-1 or IL-2 concentrations.1-3 In addition to the three types of SARS viruses, other viruses like influenza, that infect lung tissue, can induce a similar cytokine storm. Thus, treatments that resolve respiratory distress could be used in many different conditions.
A major characteristic of deteriorating lung function in COVID-19 patients is capillary leak,12 which is often the result of inflammation driven by several key inflammatory cytokines: TNF, IL-1, IL-6 and vascular endothelial growth factor (VEGF). Reactive oxygen species probably also contribute. Steroids can be used to temper this condition with limited effect, as was recently reported in the case of US President Donald Trump.
Some researchers have turned their attention to blocking cytokines to treat COVID-19, but there has been limited success so far blocking IL-6.4,5 This could be because these other cytokines are downstream from TNF, which orchestrates the many other pro-inflammatory factors that cause damage in a cytokine storm.10,13
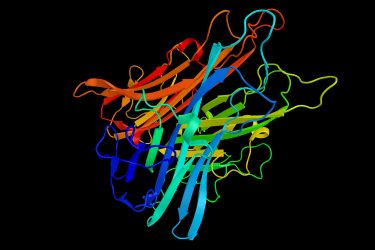
Chemical structure of tumour necrosis factor (TNF)
It may be more effective to go to the root cause of the cytokine storm, instead of blocking a piece of it. Therefore, focusing on TNF may be a better way to treat COVID-19 patients. Through the treatment of patients with various autoimmune diseases, researchers know that anti-TNF therapy reduces not only TNF, but also IL-1, IL-6, GM-CSF, VEGF and other inflammatory cytokines, which are all part of a process termed the TNF-dependent cytokine cascade, first demonstrated in rheumatoid synovial cultures.14 In sepsis,15,16 and importantly in anti-TNF treated patients,17 the efficacy of anti-TNF therapy is well documented and has resulted in TNF inhibitors becoming the world’s largest drug class since 2012.
As the inflammatory cytokines blocked by anti-TNF therapy are also present in patients with COVID-19, researchers believe that anti-TNF therapy could reduce the capillary leak present in COVID-19 patients.18,19 Consequently, anti-TNF might reduce the need for oxygen, ventilation and reduce the mortality rate associated with COVID-19.
Early observational clinical data support the potential of anti-TNF therapies in treating COVID-19; when patients with inflammatory bowel disease develop COVID-19, those undergoing anti-TNF therapy fare just as well, if not better, than those using alternative agents. In fact, anti-TNF therapy is inversely associated with COVID-19 deaths and hospital admissions.20 In patients with arthritis, those already on anti-TNF treatment do very well, having only 30 percent of the likelihood of hospitalisation compared to those not on anti-TNF.21
A track record of success
Inflammation is present in a range of diseases, including autoimmune diseases and cancer, and anti-TNF therapeutics have a broad range of applications. TNF blockers have been used for more than two decades to treat severe cases of autoimmune inflammatory disease, such as rheumatoid arthritis, inflammatory bowel disease, ankylosing spondylitis and psoriasis.22
Since it was introduced in the 1990s, anti-TNF therapy continues to be remarkably successful and is still the world’s best-selling drug class. However, only about half of patients treated with TNF blockers respond positively to therapy and more than a quarter of patients fail therapy after a year, suggesting improvements are needed.23
A problem that requires precision
The first step in improving TNF blockers is to understand the importance of the two TNF receptors: the pro-inflammatory TNFR1 and the anti-inflammatory TNFR2. Blocking TNFR2 can leave the immune system vulnerable and increase the risk of opportunistic infections, as well as respiratory and cardiac disorders. An opportunity for improvement is engineering anti-TNF therapeutics to block TNFR1 alone to preserve immune system function. However, there are currently no antibodies able to inhibit TNFR1 due to technical challenges.
We have been developing two TNF-blocking therapeutic candidates for acute or chronic inflammation: EN1001 and EN3001. EN1001 is a TNFR1-binding protein, able to inhibit TNFR1 without compromising TNFR2’s healing capabilities, while EN3001 inhibits TNFR2, useful for blocking regulatory T cells in cancer or fibrosis.
Future opportunities
There are millions of patients who could benefit from anti-TNF therapies. Autoimmune diseases, of which there are more than 80, are just one potential application. The National Stem Cell Foundation estimates that approximately four percent of the world suffers from some form of autoimmune disease. In the US, autoimmune diseases affect about 24 million people, according to the US National Institute of Environmental Health Sciences (NIEHS).
It may be more effective to go to the root cause of the cytokine storm, instead of blocking a piece of it. Therefore, focusing on TNF may be a better way to treat COVID-19 patients…
Considering the millions of people who have already been diagnosed with COVID-19 and assuming many more are still to be diagnosed, there is a great demand for effective TNF blockers. Based on an understanding of the TNF pathways, it is likely that blocking TNFR1 with EN1001 will be more effective than just blocking TNF, as it leaves regulatory T cell functioning.24,25 There is evidence that insufficient regulatory function is involved in COVID-19.26
Many believe that COVID-19 will one day be “defeated”, but unfortunately it is difficult to say the same for cancer and autoimmune diseases. The ever-present market demand coupled with the opportunity to solve one of humanity’s greatest medical challenges in recent memory – COVID-19 – should be enough of an incentive for anyone.
For two decades now, researchers have known the efficacy of anti-TNF therapies in reducing inflammation. There is still a long way to go before we truly understand COVID-19, but we know enough to know that inflammation is a big part of the equation and should consider TNF blockers as candidates for treating patients who have the coronavirus.
About the authors

Sir Marc Feldmann is Co-founder and Board Member of Enosi Life Sciences. He is a preeminent immunologist and an Emeritus Professor at the University of Oxford. With Sir Ravinder Maini, he identified TNF as a target. They led the successful trials of the first anti-TNF antibody called Infliximab (or Remicade) and prompted J&J’s $4.9 billion acquisition of Centocor. Total Remicade sales so far amount to more than $50 billion globally. Marc is a Fellow of the Royal Society of Australian Academy and a Foreign Member of the US National Academy of Sciences; he was knighted in 2010 and received the Australian equivalent. He has received many accolades including the Albert Lasker Award, the Crafoord Prize, the Canada Gairdner Award, the Paul Janssen Award and the Ernst Schering Award.

Dr H Michael Shepard is a serial entrepreneur and also serves as Chief Executive Officer, Chief Scientific Officer, Co-founder and Board Member of Enosi Life Sciences. He led the research at Genentech that resulted in Herceptin/trastuzumab, an antibody therapy now used for HER2-positive breast and gastric cancers. About three million women have been treated with Herceptin, many of them very successfully. Michael has been recognised by his peers and the Harvard Medical School Warren Alpert Prize (2006) and by the Lasker~DeBakey Clinical Medical Research Award.

Dr Fiona McCann is associate scientific director of CannBioRx Life Sciences and advisor for Enosi Life Sciences. She has previously held the position of group leader at Kennedy Institute of Rheumatology, University of Oxford, leading translational discovery programmes for autoinflammatory and fibrotic diseases. She identified a key immunoregulatory role for TNFR2 signaling in resolution of arthritis, implicating selective TNFR1 targeting as an improved therapy in chronic inflammatory disease.
References
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395(10223): 497-506.
- Del Valle DM, Kim-Schulze S, Huang HH, et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat Med 2020.
- Mann ER, Menon M, Knight SB, et al. Longitudinal immune profiling reveals key myeloid signatures associated with COVID-19. Sci Immunol 2020; 5(51).
- Della-Torre E, Campochiaro C, Cavalli G, et al. Interleukin-6 blockade with sarilumab in severe COVID-19 pneumonia with systemic hyperinflammation: an open-label cohort study. Ann Rheum Dis 2020; 79(10): 1277-85.
- Mullard A. Anti-IL-6Rs falter in COVID-19. Nat Rev Drug Discov 2020; 19(9): 577.
- Gerriets V, Bansal P, Khaddour K. Tumor Necrosis Factor (TNF) Inhibitors. StatPearls. Treasure Island (FL); 2020.
- Feldmann M, Maini RN, Woody JN, et al. Trials of anti-tumour necrosis factor therapy for COVID-19 are urgently needed. Lancet 2020.
- Hadjadj J, Yatim N, Barnabei L, et al. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science 2020; 369(6504): 718-24.
- Zhang Q, Bastard P, Liu Z, et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science 2020.
- Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev 2012; 76(1): 16-32.
- Hanley B, Naresh KN, Roufosse C, et al. Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: a post-mortem study. Lancet Microbe 2020; 1(6): e245-e53.
- Wu MA, Fossali T, Pandolfi L, et al. COVID-19: the key role of pulmonary capillary leakage. An observational cohort study. medRxiv 2020.
- Steeland S, Libert C, Vandenbroucke RE. A New Venue of TNF Targeting. Int J Mol Sci 2018; 19(5).
- Brennan FM, Chantry D, Jackson A, Maini R, Feldmann M. Inhibitory effect of TNF alpha antibodies on synovial cell interleukin-1 production in rheumatoid arthritis. Lancet 1989; 2(8657): 244-7.
- Abraham E, Wunderink R, Silverman H, et al. Efficacy and safety of monoclonal antibody to human tumor necrosis factor alpha in patients with sepsis syndrome. A randomized, controlled, double-blind, multicenter clinical trial. TNF-alpha MAb Sepsis Study Group. JAMA 1995; 273(12): 934-41.
- Cohen J, Carlet J. INTERSEPT: an international, multicenter, placebo-controlled trial of monoclonal antibody to human tumor necrosis factor-alpha in patients with sepsis. International Sepsis Trial Study Group. Crit Care Med 1996; 24(9): 1431-40.
- Feldmann M, Maini RN. Anti-TNF alpha therapy of rheumatoid arthritis: what have we learned? Annu Rev Immunol 2001; 19: 163-96.
- Charles P, Elliott MJ, Davis D, et al. Regulation of cytokines, cytokine inhibitors, and acute-phase proteins following anti-TNF-alpha therapy in rheumatoid arthritis. J Immunol 1999; 163(3): 1521-8.
- Paleolog EM, Young S, Stark AC, McCloskey RV, Feldmann M, Maini RN. Modulation of angiogenic vascular endothelial growth factor by tumor necrosis factor alpha and interleukin-1 in rheumatoid arthritis. Arthritis Rheum 1998; 41(7): 1258-65.
- Brenner EJ, Ungaro RC, Colombel JF, Kappelman MD. SECURE-IBD Database. 2020.
- Gianfrancesco M, Hyrich KL, Al-Adely S, et al. Characteristics associated with hospitalisation for COVID-19 in people with rheumatic disease: data from the COVID-19 Global Rheumatology Alliance physician-reported registry. Ann Rheum Dis 2020; 79(7): 859-66.
- Feldmann M, Maini RN. Lasker Clinical Medical Research Award. TNF defined as a therapeutic target for rheumatoid arthritis and other autoimmune diseases. Nat Med 2003; 9(10): 1245-50.
- Hamann PDH, Pauling JD, McHugh N, Hyrich K, Shaddick G, Group B-RC. Early response to anti-TNF predicts long-term outcomes including sustained remission: an analysis of the BSRBR-RA. Rheumatology (Oxford) 2020; 59(7): 1709-14.
- McCann FE, Perocheau DP, Ruspi G, et al. Selective tumor necrosis factor receptor I blockade is antiinflammatory and reveals immunoregulatory role of tumor necrosis factor receptor II in collagen-induced arthritis. Arthritis Rheumatol 2014; 66(10): 2728-38.
- Tseng WY, Huang YS, Clanchy F, et al. TNF receptor 2 signaling prevents DNA methylation at the Foxp3 promoter and prevents pathogenic conversion of regulatory T cells. Proc Natl Acad Sci U S A 2019; 116(43): 21666-72.
- Meckiff BJ, Ramírez-Suástegui C, Fajardo V, et al. Imbalance of regulatory and cytotoxic SARS-CoV-2-reactive CD4+ T cells in COVID-19. Cell 2020.
Related topics
Cytokines, Drug Repurposing, Drug Targets, Immunology, Therapeutics
Related conditions
Arthritis, autoimmune diseases, Cancer, Coronavirus, Covid-19, Cytokine storm, Inflammatory bowel disease (IBD), Psoriasis, Sepsis
Related organisations
AstraZeneca (AZ), GlaxoSmithKline (GSK), Johnson & Johnson, Merck, Moderna, Pfizer, Sanofi







