Researchers generate organoid model for gastroesophageal junction cancer
Posted: 30 December 2022 | Ria Kakkad (Drug Target Review) | No comments yet
The scientists discovered that dual knockout of genes in organoids grown from human tissue can generate a model of a potential therapeutic target for gastroesophageal junction cancer.

Researchers at Johns Hopkins Medicine, US, have created a laboratory-grown three-dimensional organoid model that is derived from human tissue and designed to advance understanding about how early stages of gastroesophageal junction (GOJ) cancer — the point where the digestive system’s food tube meets the stomach.
A report on the organoid model findings, published in Science Translational Medicine, also reveals a possible biological target for treating GOJ cancers with a drug that the researchers have already shown can slow down or stop growth of such tumours in mice.
According to the American Cancer Society, gastroesophageal cancers claim more than a million lives every year worldwide, with rates of GOJ cancer increasing more than twofold in recent decades, from 500,000 to 1 million new cases annually. Acid reflux, smoking and Helicobacter pylori bacterial infection of the stomach are well-established risk factors for tumours of the oesophagus and stomach. But experts say it has been difficult to show how cancer begins at the junction of the stomach and the oesophagus, in part due to a lack of biologically relevant GOJ-specific early disease models for research.
“Because we do not have a unique model that distinguishes GOJ tumours, gastroesophageal cancers often are classified as either oesophageal cancer or gastric cancer — not GOJ cancer,” said gastroenterologist Dr Stephen Meltzer, a corresponding author of the study. “Our model not only helps identify crucial changes happening during tumour growth at the GOJ, but also establishes a strategy for future studies to help understand tumours of other organs.”
Meltzer and a team of experts in cell biology, epigenomics, lipid profiling and big data analysis created the GOJ disease model by taking normal human biopsy tissue from patients receiving upper endoscopies. Organoids comprise 3D collections of cells derived from stem cells that can replicate characteristics of an organ or what an organ does, such as making specific kinds of cells.
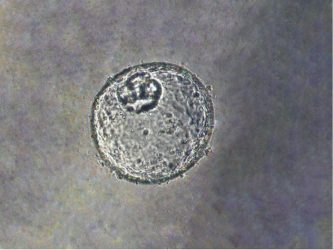

Image of human gastroesophageal junction-derived organoid, modified by dual-knockout of key tumour suppressor genes (TP53/CDKN2A) using CRISPR-Cas9 gene editing technology, which caused cells to become more cancerous. [Credit: Dr Stephen Meltzer].
Using CRISPR-Cas9, the researchers then knocked out two key tumour suppressor genes (TP53 and CDKN2A) in the organoids. Dual knockout of these genes caused cells to become more cancerous, with more rapid growth and microscopic features closer to malignancy. These altered organoids also formed tumours in immunodeficient mice.
The team further found abnormalities in a class of molecules (lipids) that store energy but also exert a variety of other functions and identified platelet activating factor as a key upregulated lipid in GOJ organoids. Platelets circulate in the bloodstream and bind together or clot when they recognize damaged blood vessels, and they can cause clotting diseases in some people. Researchers used WEB2086, which stopped the growth of implanted GOJ organoid tumours. WEB2086, a compound approved by the US Food and Drug Administration and used to treat platelet diseases, inhibits platelet activating factor receptors in mine.
Meltzer says more pre-clinical studies may be needed before using the compound for human patients, but that organoids may help advance such studies.
“Combining organoids with this gene editing method is a potentially fruitful strategy for studying other human tumours in general,” concluded Meltzer.
Related topics
CRISPR, Disease Research, Genome Editing, Genomics, Lipidomics, Oncology, Organoids, Therapeutics
Related conditions
gastroesophageal junction (GOJ) cancer
Related organisations
Johns Hopkins Medicine
Related people
Dr Stephen Meltzer