Mechanism behind Piezo proteins discovered using imaging technique
Posted: 22 August 2019 | Victoria Rees (Drug Target Review) | No comments yet
A new study has used a combination of imaging methods to understand how the Piezo protein works, improving research into treatments for conditions such as high blood pressure.
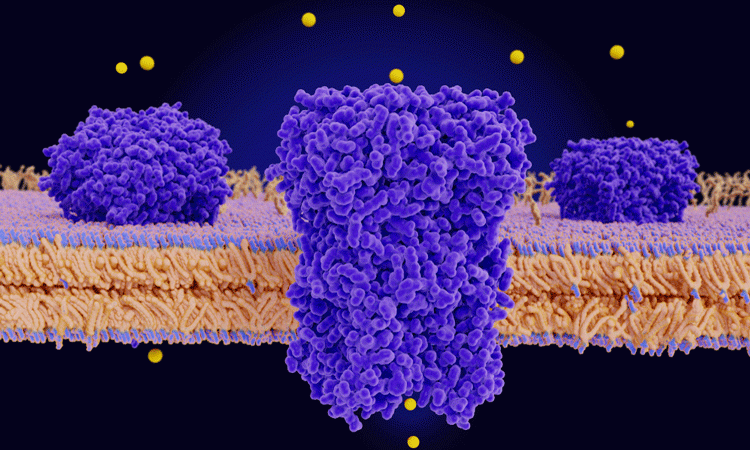
A research team has discovered the mechanism behind Piezo proteins, which function as sensors in the body for stimuli such as blood pressure. According to the authors of the study, their findings could lead to better understanding of human diseases and improve the development of new therapeutic strategies.
Researchers from Weill Cornell Medicine and The Rockefeller University, both in the US, imaged the protein, confirming its complex structure and showing how it can convert stimuli into an electrical signal.
Piezo1 and is embedded in the membranes of certain cell types and its function is to transduce mechanical force on cells into electrical signals that alter cell activity. Previous studies have demonstrated the basic structure of Piezo1, finding it has a three-armed propeller or ‘triskelion’ appearance.
From the side, it looks like a shallow bowl embedded in the cell membrane, with an ion channel at its centre. When the channel is opened, a flow of calcium and other positively charged ions into the cell are allowed through.
However, the mechanism behind the opening of the ion channel has remained unknown. Combining cryo-electron microscopy with high-speed atomic force microscopy, the team produced detailed images of Piezo1 both at rest and during the application of mechanical forces.
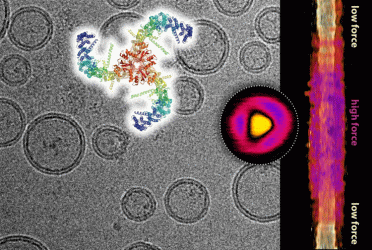
Composite of a cryo-electron microscopy image showing lipid vesicles with embedded Piezo channels and the Piezo channel structure (top). Topographic image of a single Piezo channel under force as recorded by high-speed atomic force microscopy (circle) and its lateral expansion in the membrane as a function of applied force (right) (credit: Dr Simon Scheuring).
They found that Piezo1 is a springy structure that usually bends the cell membrane where it sits but will flatten when a mechanical force is applied to the cell membrane.
“As the membrane tension increases, the structure of Piezo1 flattens and stretches out to occupy a larger area, which in turn opens the ion channel,” said co-senior author Dr Simon Scheuring, a professor at Weill Cornell Medicine.
The authors also note the possibility that other stimuli that stretch and flatten the Piezo1 structure, such as a pulling force on its arms from the inside or on an external domain called the CED from the outside the cell, could potentially open the ion channel. This would make it a suitably versatile mechanism for the wide range of cell types and physiological functions in which it works.
According to the researchers, if the membranes of cells lining blood vessels contain excess cholesterol, they would become stiffer, increasing the background tension on embedded Piezo 1 proteins and potentially disrupting the proteins’ ability to detect and help regulate blood pressure.
“Our finding leads to a great many predictions about Piezo proteins’ roles in disease that we and others can now go and investigate,” Scheuring added.
The study was published in Nature.
Related topics
Drug Targets, Imaging, Ion Channels, Research & Development
Related conditions
High blood pressure
Related organisations
Nature, The Rockefeller University, Weill Cornell Medicine
Related people
Dr Simon Scheuring