Novel method enables mapping of signalling within living cells
Posted: 22 May 2020 | Hannah Balfour (Drug Target Review) | No comments yet
A novel microscopy method has enabled researchers to study the flow of signalling information within living cells and could enhance our understanding of cancer metastasis.
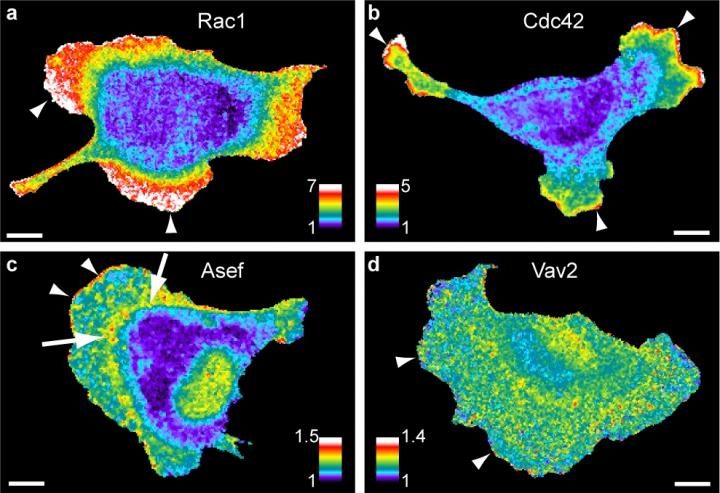

Imaging two or more protein activities at the same time allows researchers to map the signaling network coordinating cell movements. Warm colors (white, red) show sites of high activity, seen here at sites where the cell is protruding (arrowheads) [credit: Daniel Marston, PhD, UNC School of Medicine].
Why do some cancer cells move and metastasise more quickly than others? Researchers have developed a method to study the flow of signalling proteins within cells, enabling them to study how cells signal for movement and how this may change in diseases such as cancer.
“Our new tools allowed us to map the flow of signalling information within living cells and measure how much specific proteins contribute to cell behaviours, such as cell migration,” said co-first author Daniel Marston, assistant professor in the University of North Carolina (UNC) Department of Pharmacology at the UNC School of Medicine, US.
Marston and colleagues used microscopy tools developed in the lab of Klaus Hahn, the Ronald G. Thurman Distinguished Professor of Pharmacology at UNC Department of Pharmacology. A combination of these microscopy tools and fluorescent biosensors allowed the researchers to visualise the activity of multiple proteins in living cells at the same time.
Mathematical analysis methods developed by researchers at the University of Texas Southwestern Medical Center were then used by the team to quantify how proteins regulate each other.
According to the authors, the combination provides precise information about how signalling networks are wired and how they regulate cellular processes, such as cell migration and metastasis.
The team suggest this method will enable them to understand how networks operate in health and disease and compare data from healthy cells to cell movement during various health conditions, such as inflammation, a hallmark of many diseases.
“If we can find out how these movement processes are changed in diseases, such as cancers, then we might be able to design better treatments effective only for that particular disease, while keeping other cells healthy,” Marston added.
The paper was published in Nature Chemical Biology.
Related topics
Analytical Techniques, Disease Research, Imaging, Oncology, Protein, Proteomics, Technology
Related conditions
Cancer, metastatic cancer
Related organisations
University of North Carolina (UNC), University of Texas Southwestern Medical Center
Related people
Daniel Marston, Klaus Hahn