How will CRISPR evolve in the future?
Posted: 26 November 2019 | Victoria Rees (Drug Target Review) | No comments yet
CRISPR is a tool used by researchers to precisely edit genes and has shown potential for treating genetic diseases. This article delves into some recent developments and explores what the future holds for CRISPR.

CRISPR genome editing is a promising field that enables researchers to precisely delete, replace or edit genes.
CRISPR-Cas is a prokaryotic defence system whereby bacteria use RNA molecules and CRISPR-associated (Cas) proteins to target and destroy the DNA of invading viruses. This molecular machinery has been repurposed by researchers to target and edit specific sections of any DNA, whether bacterial or human.
Challenges of CRISPR technology
Despite the success of CRISPR, the technique is far from refined. In certain situations, the editing process can result in off-target DNA being changed, causing unwanted effects. Also, CRISPR-Cas9 is a large molecular complex, with both the Cas9 nuclease and an engineered single-guide RNA (sgRNA) that helps the nuclease locate its target. This can make its delivery into the nucleus of the cell, where CRISPR needs to access DNA, difficult.
Consequently, many researchers have sought improvements to CRISPR with the gene editing method expected to continue development well into the future.
Here, three researcher groups who have contributed to recent CRISPR developments explain their work and predict how CRISPR may evolve.
Simultaneous edits
In an attempt to multiplex CRISPR systems to target lots of genes, researchers at ETH Zurich in Switzerland swapped the Cas9 enzyme for Cas12a. Using this plasmid allowed the researchers to simultaneously edit genes in 25 target sites. The team predicts that dozens or even hundreds more sites could be modified using this method.

Genes and proteins in cells interact in many different ways. Each dot represents a gene; the lines are their interactions. For the first time, the new method uses biotechnology to influence entire gene networks in one single step (credit: ETH Zurich/Carlo Cosimo Campa).
Cas12a enabled the researchers to attach shorter sgRNA address molecules than when using Cas9. The shorter length molecules mean that more can fit onto the plasmid, which is a circular DNA molecule that acts as the blueprint of the Cas enzyme, thus enabling CRISPR to edit many genes in a short space of time.
Professor Randall Platt, who led the research, explained that his team’s technique is “conditional, inducible and orthogonal.”
This development offers an improvement on traditional CRISPR technology, which only enables one gene to be edited at a time. This technique therefore speeds the process up, allowing CRISPR to edit many genes simultaneously. It also means that the expression of some genes can upregulated while others can be downregulated.
Platt says that their technique is “drastically better,” at targeting multiple genes and it afforded the researchers “sophisticated control over cellular genomes and transcriptomes.”
Multiple protein approach
Another development for CRISPR technologies came from researchers at Duke University in the US. The team successfully used Class 1 CRISPR systems for the first time to edit the epigenome of human cells. Conventional CRISPR-Cas9 methods are categorised as Class 2 systems.
The Class 1 technique makes use of multiple proteins in a process called CRISPR-associated complex for antiviral defence (Cascade). This complex binds with high accuracy to the correct sites. After binding, Cascade utilises a Cas3 protein to target and edit the DNA. They were also able to both activate and repress target gene expression.
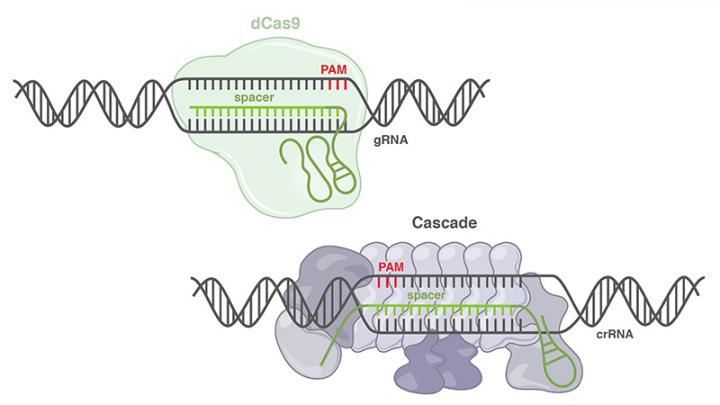
Illustrations representing the components of the common dCas9 system (top) and the Cascade system (bottom) (credit: Gersbach Lab).
The team says that this research contributes to an enhancement of CRISPR technologies as it provides a potential alternative for CRISPR-Cas9 when there are complications such as immune responses to Cas proteins. It can also recruit various modifiers of gene regulation, including activators and repressors, to a gene.
Associate Professor Charles Gersbach, one of the lead researchers, says that the team will continue to explore CRISPR biology and how the Class 1 method can be developed for gene editing.
“It will be exciting to explore other types of effector domains, such as modifiers of DNA methylation, base editors, etc, attached to Cascade,” Gersbach says.
Delivery systems
A further CRISPR development has come from a collaboration between Tufts University in the US and the Chinese Academy of Sciences. These researchers used a biodegradable synthetic lipid nanoparticle to deliver their CRISPR editing tools into the cell to precisely alter the cells’ genetic code.
According to the team, their method resulted in up to 90 percent efficacy in gene editing. The lipid nanoparticles encapsulate messenger RNA (mRNA) encoding Cas9. Once the contents of the nanoparticles – including the sgRNA – are released into the cell, the cell’s protein-making machinery takes over and creates Cas9 from the mRNA template.
A unique feature of the nanoparticles is made of synthetic lipids comprising disulfide bonds in the fatty chain. When the particles enter the cell, the environment within the cell breaks open the disulfide bond to disassemble the nanoparticles and the contents are quickly and efficiently released into the cell.
Once the contents of the nanoparticles… are released into the cell, the cell’s protein-making machinery takes over and creates Cas9″
The researchers highlighted that their delivery system refines CRISPR technologies; as Cas9 is a large complex it is difficult to deposit directly into the nucleus of the cell. Other research teams have used viruses, polymers and other kinds of nanoparticles to deliver CRISPR-Cas9, but the low efficiency of transfer limits its success. As their delivery system builds the Cas9 enzyme later in the process, it has high levels of transfer and efficacy.
Professor Qiaobing Xu, a co-corresponding author of the study, highlighted that the synthetic lipid could be made with low-toxicity. Furthermore, he explained, as there is no limit in terms of cargo size, the lipid is an improvement upon viral delivery.
He also emphasised that with viral delivery, there is always a concern about the immune response against the viral particle, but a non-viral delivery method does not have this disadvantage.
How will CRISPR improve?
These developments in the CRISPR technique indicate how the technology is set to improve and develop in the future. However, research is far from over.
Platt believes that CRISPR processes are still in their infancy, as the current tools are effective at cutting DNA but can result in random repair. He believes that the future of genome editing is going to require new tools to enable more precise changes to the genome.
 Eliminating random output would ensure success of the technology for therapeutic effect. Making precise changes is therefore the direction that CRISPR will evolve to, allowing more complex challenges to be tackled.
Eliminating random output would ensure success of the technology for therapeutic effect. Making precise changes is therefore the direction that CRISPR will evolve to, allowing more complex challenges to be tackled.
Gersbach remarks that his team’s study will likely stimulate more research into Class 1 systems, which could lead to numerous applications and provide more biological insights into its potential therapeutic use.
Although there is more work to be done with regard to Class 1 CRISPR systems, its unique attributes make it worth investigating, he says.
Xu also comments that CRISPR is a young field compared with other technologies. He highlights the many areas of CRISPR developments: better editors; larger animal or in vitro models; and more precise analytical methods to detect gene editing.
He believes that CRISPR holds tremendous potential to treat disease, which is “absolutely ground-breaking.” If specific, targeted genes in the body can be controlled, then almost every condition could potentially be treated.
Summary
In conclusion, CRISPR can be a highly useful tool for editing genes and to potentially treat complex diseases. However, it still must be refined as a technique. This has caused researchers to strive for improvements in this area, to make the process more precise and effective.
These recent studies demonstrate that improvements are possible and serve to highlight the enormous potential that CRISPR offers.
According to the researchers, CRISPR technologies have progressed and will continue to improve. They all agree that CRISPR could one day be an effective way to treat genetic diseases.
Related topics
CRISPR, DNA, Drug Delivery, Drug Targets, Gene Therapy, Genome Editing, Research & Development, Targets
Related organisations
Chinese Academy of Sciences, Duke University, ETH Zurich, Tufts University







