Incorporating microglia into brain organoids
By  Drug Target Review2023-11-16T16:00:30
Drug Target Review2023-11-16T16:00:30
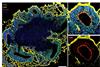
iPS-cell-derived microglia in brain organoids have enabled scientists to understand early brain development and microglia-associated disease.
Already a member? Sign in
Intelligence for the future of drug discovery
By highlighting cutting-edge research and its practical applications, Drug Target Review enables readers to make decisions that accelerate innovation, translate discoveries into therapies, and create tangible benefits for patients worldwide.
Become a member to continue reading
By becoming a member you join a connected and collaborative community driving the industry forward. Member benefits include:
- Unlimited access to industry intelligence that can make a difference to your business
- A weekly newsletter keeping the most vital information at the front of your thinking
- Exclusive invitations to powerful webinars and events featuring high profile experts
- Access to deep-dive reports and analysis that reveal exciting new opportunities for business growth and transformation
Related articles
Partner content
-

-
 Whitepaper
WhitepaperPoster: Antibodies for microglial activation states
2023-01-05T14:41:33
Sponsored by Cell Signaling Technology
-
 Video
VideoVideo: Spatial imaging as a tool for discovery
2023-03-02T13:57:58
Sponsored by Cell Signaling Technology
-
 Whitepaper
WhitepaperCase Study: Target discovery for rheumatoid arthritis
2024-01-19T07:40:55
Sponsored by Euretos
-
 Whitepaper
WhitepaperApplication note: In situ identification of cancer stem cell niches (HCC)
2024-02-01T11:52:50
Sponsored by Leica Microsystems
-
 Whitepaper
WhitepaperApplication note: Enrichment of high titer CHO clones
2023-03-03T14:06:14
Sponsored by Bio-Techne
- Topics
- Technology
- Resources
- Diseases
- Drugs and therapies
- Research and development
- Tools and techniques
- Business
- Advertising & Editorial
- Advertising opportunities
- Media planner
- Advertising tech specs
- Information for authors
- Reuse policy
- Information
- About us
- Company information
- Careers
- Terms and conditions
- Privacy policy
- Cookie policy
- Topics A-Z
- Writers A-Z
- © 2026 Drug Target Review
Site powered by Webvision Cloud









