Protein responsible for muscle degeneration identified
Posted: 2 October 2019 | Victoria Rees (Drug Target Review) | No comments yet
A study has demonstrated that a certain protein is critical in the decline of muscle regeneration, with the researchers also showing how to inhibit this process in mice.
Researchers have discovered a protein which suppresses the ability of aged muscle stem cells to regenerate. They indicate that their findings reveal a potential therapeutic pathway for muscle degeneration in the elderly.
The study was conducted at the Carnegie Institution for Science, US. Previous studies have demonstrated that skeletal muscles have the capacity to make new muscles from stem cells, which can self-renew. However, this diminishes with age, resulting in poorer muscle regeneration from muscle trauma.
Using mouse models, the team found that the GAS1 protein is responsible for this age-related decline, which is encoded by the growth-arrest specific gene. They also found that the protein is found in only a small number of young muscle stem cells but is present in all aged muscle stem cells.
Editing the mice genes, the researchers heightened GAS1 expression in the young stem cell population, causing diminished regeneration. By contrast, removing GAS1 from the aged muscle stem cells rejuvenated them to a youthful state which supported regeneration.
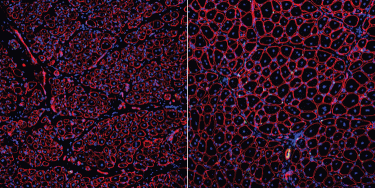
These two photographs show how treatment with the binding-protein GDNF repairs aged muscle stem cells to support robust muscle regeneration. The image on the left shows small muscle fibers (red shapes with blue nuclei inside), which were made by old muscle stem cells. The image on the right shows increased muscle fiber size and muscle stem cell numbers (indicated by green dots) due to GDNF treatment (credit: Liangji Li).
A further discovery of the team is that GAS1 inhibits another protein, a cell-surface receptor called RET, which they showed to be necessary for muscle stem cell renewal. The more GAS1 protein present, the more RET function was reduced.
A third protein called GDNF was able to reverse this inhibition of RET. When the researchers injected GDNF to the muscles of aged mice, muscle stem cell function and muscle regeneration were restored.
“With a rapidly ageing population, issues like muscle deterioration are an increasing societal challenge,” said Chen-Ming Fan, MD, the lead researcher. “Our work could reveal a potential pathway for therapeutic targeting to combat muscle degeneration in the elderly.”
The results were published in Nature Metabolism.
Related topics
Drug Targets, Protein, Regenerative Medicine, Research & Development, Stem Cells, Targets
Related conditions
Muscle degeneration
Related organisations
Carnegie Institution for Science
Related people
Chen-Ming Fan