‘Cell-projection pumping’ presents new cancer target, say researchers
Posted: 27 March 2020 | Victoria Rees (Drug Target Review) | No comments yet
A new discovery about how cancer cells exchange contents with fibroblasts presents a new oncologic drug target, according to the researchers.

A novel finding by researchers has revealed a new source of cancer cell diversity, which the scientists say could have profound implications for cancer treatment.
Although most cancer cells are killed by chemotherapy, individual cells vary in their sensitivity, so that some cancer cells often escape. Therefore, diversity among cancer cells is an issue for cancer treatment.
Previously diversity was thought to be mostly due to genetic variability among cancer cells. Now, however, researchers at the University of Sydney, Australia, have discovered a whole new source of cancer cell diversity.
In 2012, a team led by Professor Hans Zoellner from the University of Sydney School of Dentistry, found that cancer cells exchange contents with surrounding normal cells called fibroblasts, but the mechanism remained unknown, until now.
All cells constantly probe each other with tentacle-like cell-projections. These reach out to neighbours, probe and then retract. When a cell-projection retracts, there is a brief increase in fluid pressure within the projection, to force cytoplasm back into the cell body.
Studying cells in time-lapse films, Zoellner noticed that transfer is from retracting cell-projections. He reasoned that transient micro-fusions between the retracting cell-projections and any neighbouring cell would permit cytoplasm from the cell-projection to be injected into the neighbour, instead of being returned to the fibroblast.
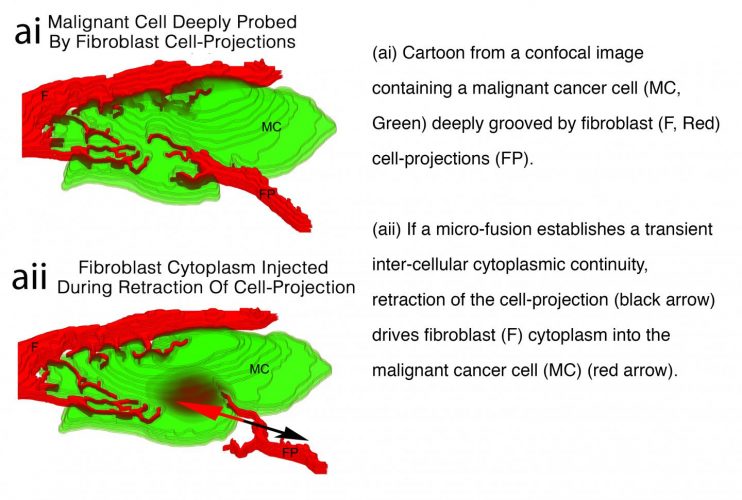

Illustration of malignant cell deeply probed by fibroblast cell projections [credit: Prof Hans Zoellner/University of Sydney].
Zoellner and his team tested this idea by combining mathematical modelling, cell experimentation and computer simulation. They named their outcome ‘cell-projection pumping’.
The significance of cell-projection pumping is underscored by separate work published by Professor Zoellner’s team. They found that cancer cells become more diverse in size and shape and also migrate more quickly after cell-projection pumping.
“This is a whole new cancer target. Now that we know it’s happening, we can think about trying to block it and work towards better outcomes,” said Zoellner.
The results are published in Biophysical Journal.
Related topics
Disease Research, Drug Targets, Oncology, Research & Development, Targets
Related conditions
Cancer
Related organisations
Sydney University
Related people
Professor Hans Zoellner








