Genetic deletion permanently prevented type 1 diabetes development in mice
Posted: 30 March 2020 | Hannah Balfour (Drug Target Review) | No comments yet
Researchers observed that deleting the IRE1-alpha gene caused beta cells to de-differentiate and then re-differentiate in mice, preventing immune system auto-activation.


Scientists found that de-differentiating pancreatic beta cells, the insulin producing cells which are targeted and destroyed by the immune system in type 1 diabetes, prevented their degradation and immune activation even once they differentiated into the mature cell type again.
According to University of Wisconsin-Madison (UW-Madison), US, researcher, Feyza Engin, a biomolecular chemistry professor at the UW-Madison and lead author of the study published in Cell Metabolism, those at high risk of developing type 1 diabetes can be identified by the presence of auto-antibodies in their blood; however, very little is currently done in clinic as there is no treatment for diabetes, other than life-time management.
To overcome this problem, Engin’s lab genetically altered a line of mice destined to develop type 1 diabetes, by removing a gene involved in the cell stress response, IRE1-alpha. According to Engin, the deletion was expected to expedite the development of diabetes; however, the researchers observed that the mice underwent an initial period of hyperglycaemia and then their blood glucose levels returned to a normal level.
She said that the cells were becoming normal insulin producers, but underwent a stage of immaturity first. “Once we remove this gene, it’s almost like the beta cells are undergoing a disguise,” said Engin. “They lose their mature identity. They de-differentiate and exhibit features of progenitor cells and express hormones of other cell types in addition to insulin.”
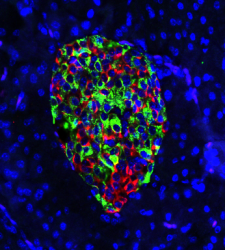

Cells in the pancreas of a gene-edited mouse produce nearly equal amounts of the hormones insulin (green) and glucagon (red). In mice that develop type 1 diabetes, green would predominate and insulin production would draw the deadly attention of a disordered immune system [credit: Hugo Lee, study first author].
The researchers observed that if the de-differentiation occurred before an auto-immune response by T cells, then the T cells responded differently permanently. Elgin stated: “When they de-differentiate, they don’t act like typical beta cells anymore. They reduce the expression of many genes that signal to immune cells… altering the diabetogenic activity of T cells. They don’t really recognise the beta cells as a problem anymore. They don’t attack.”
Importantly the immature de-differentiated beta cells re-differentiate into the mature type and go on to produce insulin as they would in non-diabetic mice. According to Elgin, the period of hyperglycaemia observed in mice is while the de-differentiation and re-differentiation occurs.
“We’ve found a very important time point where de-differentiation helps greatly reduce the immune cells’ diabetogenic activity,” Engin said. She continued that the same treatment mechanism could be used to treat other autoimmune conditions, such as arthritis, lupus and multiple sclerosis: “If you can determine an appropriate cell targeted by auto-immune response and make those victim cells less functional, less mature in the beginning, maybe they can avoid their role in the progress of other diseases too”.
Related topics
Cell-based assays, Disease Research, Drug Targets, Genome Editing, Genomics, Immunology, T cells
Related conditions
Arthritis, Lupus, Multiple Sclerosis (MS), type 1 diabetes
Related organisations
University of Wisconsin-Madison (UW-Madison)
Related people
Feyza Engin