“Organs-on-a-chip” reveal how digestive tract may influence neurological disease
Posted: 1 February 2021 | Victoria Rees (Drug Target Review) | No comments yet
A team has used their new organs-on-a-chip system to replicate the interactions between the brain, liver and colon.
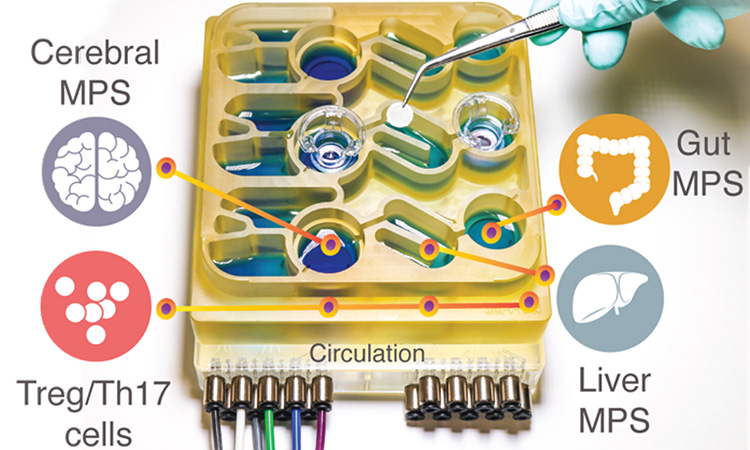
The organs-on-a-chip system [credit: Martin Trapecar/MIT].
To help researchers better understand the gut-brain axis, researchers have developed an “organs-on-a-chip” system that replicates interactions between the brain, liver and colon. The organoid system was developed at MIT, US.
Using that system, the researchers were able to model the influence that microbes living in the gut have on both healthy brain tissue and tissue samples derived from patients with Parkinson’s disease. They found that short-chain fatty acids, which are produced by microbes in the gut and are transported to the brain, can have very different effects on healthy and diseased brain cells.
“While short-chain fatty acids are largely beneficial to human health, we observed that under certain conditions they can further exacerbate certain brain pathologies, such as protein misfolding and neuronal death, related to Parkinson’s disease,” said Martin Trapecar, an MIT postdoc and lead author of the study.
For several years, the researchers have been developing microphysiological systems – small devices that can be used to grow engineered tissue models of different organs, connected by microfluidic channels. They found that short-chain fatty acids (SCFAs), molecules produced by microbes in the gut, can worsen autoimmune inflammation associated with ulcerative colitis under certain conditions. SCFAs, which include butyrate, propionate and acetate, can also have beneficial effects on tissues, including increased immune tolerance.
Previous research has revealed a connection between SCFAs and Parkinson’s disease in mice. SCFAs, which are produced by bacteria as they consume undigested fibre in the gut, sped up the progression of the disease, while mice raised in a germ-free environment are slower to develop the disease.
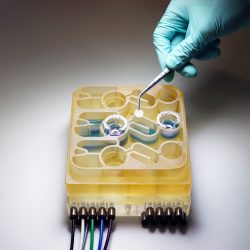
Credit: Martin Trapecar/MIT.
The team decided to further explore these findings, using their microphysiological model. In the new study, the scientists added the brain and circulating immune cells to their multiorgan system. The cells that the MIT researchers used for their Parkinson’s model carry a mutation that causes accumulation of a protein called alpha synuclein, which damages neurons and causes inflammation in brain cells. The researchers generated brain cells that have this mutation corrected but are otherwise genetically identical and from the same patient as the diseased cells.
They studied these two sets of brain cells in microphysiological systems that were not connected to any other tissues and found that the Parkinson’s cells showed more inflammation than the healthy, corrected cells. The Parkinson’s cells also had impairments in their ability to metabolise lipids and cholesterol.
The researchers then connected the brain cells to tissue models of the colon and liver, using channels that allow immune cells and nutrients, including SCFAs, to flow between them. They found that for healthy brain cells, being exposed to SCFAs is beneficial and helps them to mature. However, when brain cells derived from Parkinson’s patients were exposed to SCFAs, the beneficial effects disappeared. Instead, the cells experienced higher levels of protein misfolding and cell death. These effects were seen even when immune cells were removed from the system, leading the researchers to hypothesise that the effects are mediated by changes to lipid metabolism.
“It seems that short-chain fatty acids can be linked to neurodegenerative diseases by affecting lipid metabolism rather than directly affecting a certain immune cell population,” Trapecar said. “Now the goal for us is to try to understand this.”
The researchers also plan to model other types of neurological diseases that may be influenced by the gut microbiome using their organs-on-a-chip system. The findings offer support for the idea that human tissue models could yield information that animal models cannot. Professor Linda Griffith, one of the senior authors of the paper, is now working on a new version of the model that will include micro blood vessels connecting different tissue types, allowing researchers to study how blood flow between tissues influences them.
“We should be really pushing development of these, because it is important to start bringing more human features into our models,” Griffith said. “We have been able to start getting insights into the human condition that are hard to get from mice.”
The study on organs-on-a-chip was published in Science Advances.
Related topics
In Vitro, Organ-on-a-Chip, Organoids, Research & Development
Related conditions
Parkinson's disease (PD)
Related organisations
MIT
Related people
Martin Trapecar, Professor Linda Griffith