Researchers visualise metabolic processes at the single-cell level
Posted: 18 June 2021 | Victoria Rees (Drug Target Review) | No comments yet
Scientists have used imaging methods and machine learning to understand cellular metabolism at the single-cell level.
For the first time, researchers have visualised metabolic processes at the single-cell level. According to the team, from the University of Chicago, US, understanding cellular metabolism could be key to treating a wide array of diseases, including vascular diseases and cancer.
The researchers developed a combined imaging and machine learning technique that can measure a metabolic process at both the cellular and sub-cellular levels. Using a genetically encoded biosensor paired with artificial intelligence (AI), they were able to measure the glycolysis of single endothelial cells, which line blood vessels.
“Understanding cellular metabolism is universally important,” said Associate Professor Yun Fang, co-lead researcher of the study. “By measuring single-cell metabolism, we potentially have a new way of treating a wide range of diseases… This is the first time that we can visualise cellular metabolism at different temporal and spatial scales, even at the subcellular level, which could fundamentally change the language and approach for researchers to study cellular metabolism.”
The team explain that endothelial cells provide a tight layer inside blood vessels, but can contract when they need help from the immune system. Abnormal contraction can cause leaky blood vessels, leading to heart attack or stroke.
To better understand how cells metabolise energy to fuel this contraction, the researchers turned to Förster resonance energy transfer sensors – genetically encoded biosensors that can measure the amount of lactate inside cells. Lactate is the byproduct of glycolysis.
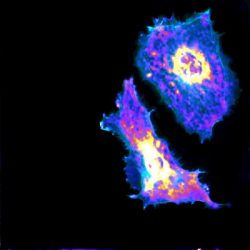
A new PME imaging and machine learning technique can measure glycolysis at both the cellular and subcellular levels, potentially leading to new methods for treating a wide array of diseases, including cancer and COVID-19 [credit: Fang Lab at University of Chicago].
Though the researchers did not create the sensors, by pairing the sensors with machine learning algorithms, they created a technique that allowed them to image cells, analyse the data and parse out glycolysis reactions at the single-cell and subcellular levels.
“Now we can look at and understand details within the cells, like certain areas of cells where there is an increase of glycolysis,” Fang said. “This is a key technological innovation.”
They were able to measure just how much glucose cells used when they contracted and moved and they also found a new mechanism of glucose transport mediated by the cell’s cytoskeleton – a receptor called GLUT3 – that these cells use to uptake glucose.
The team say that these findings have important implications in treating cancer. Endothelial migration and proliferation, driven by glycolysis, are major cellular processes involved in vascular growth, which is necessary for tumour survival and growth.
“If we can find a way to inhibit contraction, we could [also] lessen acute respiratory distress syndrome (ARDS) in COVID-19 patients,” Fang said.
The research on single-cell metabolism was published in Nature Metabolism.
Related topics
Analytical Techniques, Artificial Intelligence, Imaging, Informatics, Metabolomics
Related organisations
University of Chicago
Related people
Associate Professor Yun Fang