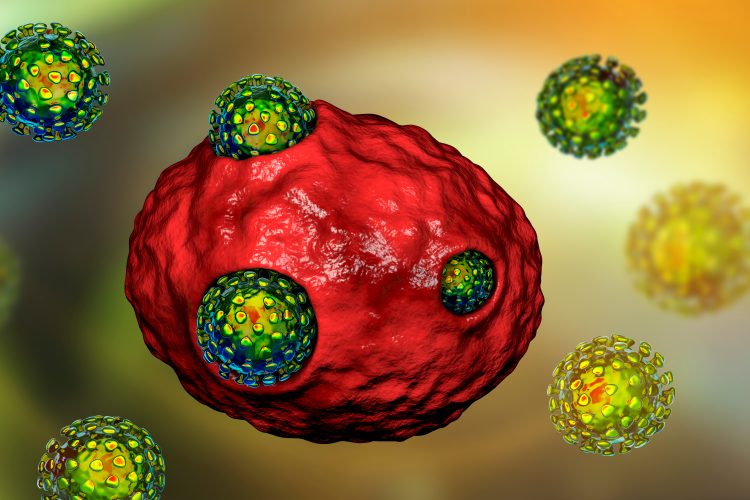
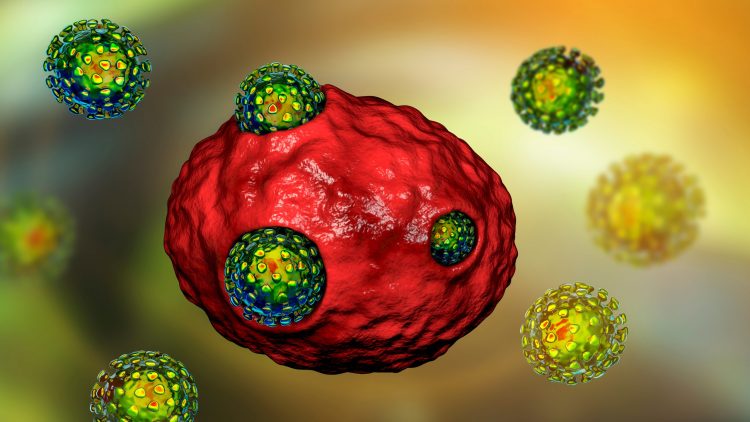
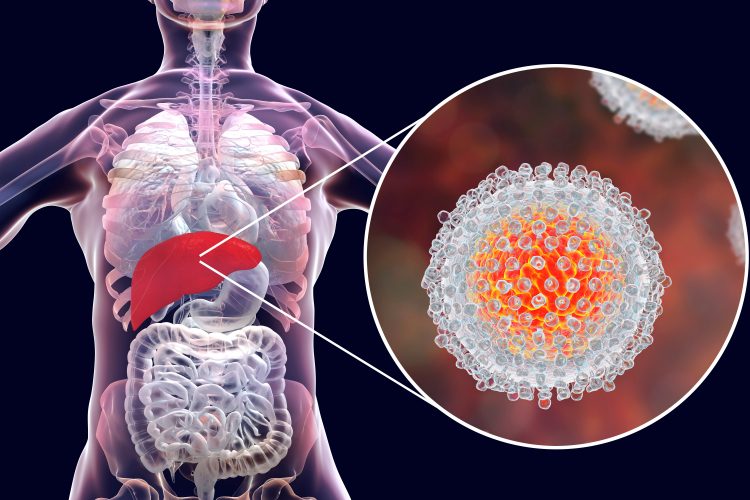
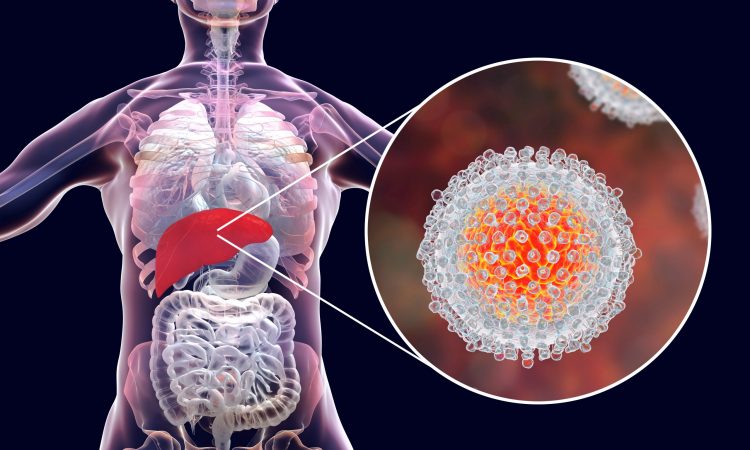
Obstacles and innovations of macrocyclic drug development
Macrocycles outperform traditional therapeutics on several fronts, yet they account for just four percent of FDA-approved drugs. Colleagues Ann E Cleves and Ajay N Jain from Optibrium Ltd explain why we can expect to see this number increase in the coming years.