Model to study oculocutaneous albinism eye conditions developed
Posted: 31 January 2022 | Victoria Rees (Drug Target Review) | No comments yet
The first patient-derived stem cell model to research eye conditions related to oculocutaneous albinism (OCA) has been created.


Researchers at the US National Eye Institute (NEI) have developed the first patient-derived stem cell model for studying eye conditions related to oculocutaneous albinism (OCA).
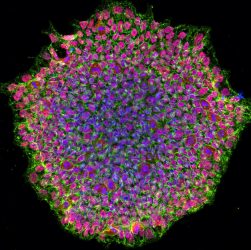

A human iPSC colony from OCA1A patient. The image was acquired using a confocal microscope and is stained for pluripotency marker proteins. The red colour depicts transcription factor OCT4, green is SSEA4 protein and blue represents the nucleus of the cells [credit: NEI].
According to the researchers, OCA is a set of genetic conditions that affects pigmentation in the eye, skin and hair due to mutation in the genes crucial to melanin pigment production. In the eye, pigment is present in the retinal pigment epithelium (RPE) and aids vision by preventing the scattering of light. The RPE is located right next to the eye’s light-sensing photoreceptors and provides them nourishment and support. People with OCA lack pigmented RPE and have an underdeveloped fovea, an area within the retina that is crucial for central vision.
People with OCA have misrouted optic nerve fibres, which carry visual signals to the brain. Scientists think that RPE plays a role in forming these structures and want to understand how lack of pigment affects their development.
“Animals used to study albinism are less than ideal because they lack foveae,” said Dr Brian Brooks, one of the authors of the paper published in Stem Cell Reports. “A human stem cell model that mimics the disease is an important step forward in understanding albinism and testing potential therapies to treat it.”
To make the model, the researchers reprogrammed skin cells from individuals without OCA and people with the two most common types of OCA (OCA1A and OCA2) into induced pluripotent stem cells (iPSCs). The iPSCs were then differentiated to RPE cells. The RPE cells from OCA patients were identical to RPE cells from unaffected individuals but displayed significantly reduced pigmentation.
The researchers say they will use the model to study how lack of pigmentation affects RPE physiology and function. In theory, if fovea development is dependent on RPE pigmentation and pigmentation can be somehow improved, vision defects associated with abnormal fovea development could be at least partially resolved.
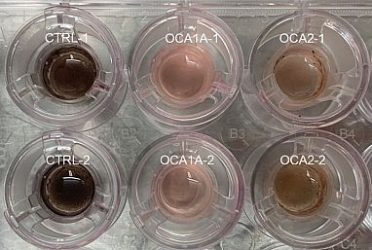

Human iPSC derived RPE monolayer on top of the membrane in a trans-well culture system. Unaffected RPE cells (CTRL1 and 2) exhibiting pigmentation and OCA1A and OCA2 RPE from albinism patients exhibiting pigmentation defects [credit: NEI].
“Treating albinism at a very young age, perhaps even prenatally, when the eye’s structures are forming, would have the greatest chance of rescuing vision,” said Brooks. “In adults, benefits might be limited to improvements in photosensitivity, for example, but children may see more dramatic effects.”
The team say they are now exploring how to use their model for high-throughput screening of potential OCA therapies.
“This ‘disease-in-a-dish’ system will help us understand how the absence of pigment in albinism leads to abnormal development of the retina, optic nerve fibres and other eye structures crucial for central vision,” said Dr Aman George, the lead author of the paper.
Related topics
Disease Research, High-Throughput Screening (HTS), Induced Pluripotent Stem Cells (iPSCs), Stem Cells
Related conditions
Oculocutaneous albinism (OCA)
Related people
Dr Aman George, Dr Brian Brooks