Researchers reveal how fungi produce potential therapeutic compounds
Posted: 23 September 2019 | Victoria Rees (Drug Target Review) | No comments yet
A team has discovered that fungi use the Diels-Alder reaction to assemble new compounds that have potential pharmaceutical uses.
A study has found how natural processes in fungi can produce large quantities of bioactive compounds. The research team say that these prenylated indole alkaloids have a wide range of potential applications as useful drugs.
The research was conducted by the University of Michigan Life Sciences, US. The genes of the fungi encode for enzymes which use very simple instructions to form complex molecules.
We gain… insight into how to use this as a tool to synthesise new chemical compounds with biological activities”
“If we can actually isolate the genes involved and make these enzymes, we should be able to recreate the entire bio-assembly line in a test tube,” said Qingyun Dan, a researcher in the lab and a lead author on the study. “But, until now, no lab has been able to do so.”
Using a collaborative approach that combined synthetic chemistry, genetics, enzymology, computational chemistry and structural biology, the researchers uncovered the process. They found that the final step in the assembly process is the Diels-Alder reaction, which is nearly unprecedented in nature.
“Even within the last few years, there has been a major debate in the field about whether this reaction actually exists in nature. It’s just the most phenomenal, unexpected path to this fascinating class of indole alkaloids,” added David Sherman, one of the senior authors of the study.
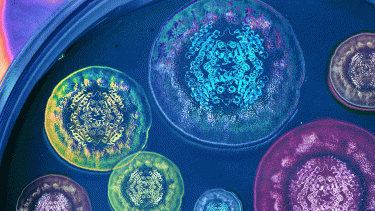
Representation of the Diels-Alderase complex structure with premalbrancheamide, one of the natural products produced by the fungi shown (credit: Life Sciences Institute multimedia designer Rajani Arora).
The team believe that discovering the enzyme that enables the Diels-Alder reaction, resolving how these compounds are made in nature, now opens two exciting paths forward.
Firstly, the particular enzyme that catalyses this Diels-Alder reaction could help improve one of the most commonly used chemical reactions. This enzyme performs the reaction with specificity greater than can be achieved in the lab, meaning it creates only the one desired compound and no unintended by-products.
Secondly, because the researchers were able to obtain a crystal structure of the enzyme performing the Diels-Alder reaction, they now have a clear picture of how the enzyme directs the reaction in nature – and how it might be harnessed to create new compounds in the future.
“This is a very good example of the explanatory power of crystal structures,” said Sean Newmister, a researcher and a lead author on the study. “We gain mechanistic insight into the activity of the enzyme we’re studying, but also insight into how to use this as a tool to synthesise new chemical compounds with biological activities. And that’s really exciting.”
The results are published in Nature Chemistry.
Related topics
Biopharmaceuticals, Drug Discovery, Drug Targets, Enzymes, Molecular Targets, Research & Development, Screening
Related organisations
University of Michigan Life Sciences Institute
Related people
David Sherman, Qingyun Dan, Sean Newmister