New gene therapy for Huntington’s demonstrates success in mice
Posted: 2 March 2020 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have developed a new regenerative gene therapy using neurogenic differentiation, which has shown efficacy treating Huntington’s disease in mice.
A new gene therapy to potentially treat Huntington’s disease has regenerated the functional neurons in mouse models of the condition. The research was conducted at Penn State University, US, and Jinan University, China.
“We are developing a series of neurogenic differentiation 1 (NeuroD1) based gene therapies to reprogramme brain internal glial cells directly into functional new neurons to treat a variety of brain disorders including Huntington’s Disease, Alzheimer’s disease, stroke, amyotrophic lateral sclerosis (ALS) and many more,” said lead researcher Dr Gong Chen. “Because every single neuron in our brain is surrounded by supporting glial cells, such direct glia-to-neuron conversion technology offers great advantages over stem cell transplantation therapy in terms of high efficiency of neuroregeneration and no worries about immunorejection.”
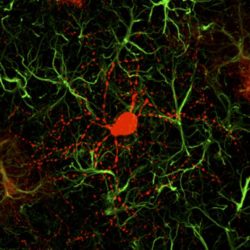
This is a newly converted neuron (red) surrounded by astrocytes (green) [credit: Gong Chen and Zheng Wu].
NeuroD1 promotes neuronal generation during normal brain development. Chen’s team has previously demonstrated that expression of NeuroD1 in the mouse brain can directly convert cortical astrocytes (one subtype of glial cells) into functional neurons.
The team has also revealed that NeuroD1-generated neurons are mainly glutamatergic neurons, which are the major driving force of brain activity. However, Huntington’s is caused by the degeneration of GABAergic neurons, a type of inhibitory neuron that accounts for more than 90 percent of the neurons in the striatum region. In order to generate GABAergic neurons, the researchers combined NeuroD1 together with another transcription factor Dlx2, known to generate GABAergic neurons during early brain development and successfully converted striatal astrocytes into GABAergic neurons in mice with Huntington’s. The scientists note that they used adeno-associated virus (AAV) vectors to develop their gene therapy.
In their mouse experiments, Dr Chen and colleagues reported that 80 percent of the AAV-infected striatal astrocytes were directly converted into GABAergic neurons and the remaining astrocytes could proliferate to replenish themselves. Essentially all of the newly generated neurons were electrophysiologically functional, forming synaptic connections with other neurons. They further demonstrated that the newly generated neurons can project their axons to the right target areas, suggesting that they have integrated into global brain circuits. According to the researchers, the mice treated with the gene therapy exhibited significant motor functional recovery and remarkable extension of life span.
“Our regenerative gene therapy approach is different from conventional gene therapy that typically aims at the mutant genes by either correcting the gene mutations or reducing the mutant gene product, such as reducing mHtt aggregates in Huntington’s patients,” Dr Chen said. “Obviously, reducing mHtt aggregates at an early stage might slowdown the disease progression but it cannot regenerate new neurons for the late stage patients. An ideal approach may be to combine our neuroregenerative approach together with gene correction technology to generate healthy new neurons in future studies,” concluded Dr Chen.
The work has been published in Nature Communications.
Related topics
Drug Targets, Gene Therapy, Genomics, Neurons, Regenerative Medicine
Related conditions
Huntington's disease
Related organisations
Jinan University, Penn State University
Related people
Dr Gong Chen