Induced pluripotent stem cells and CRISPR reversed diabetes in mice
Posted: 23 April 2020 | Victoria Rees (Drug Target Review) | No comments yet
Induced pluripotent stem cells made to produce insulin and CRISPR, used to correct a genetic defect, cured Wolfram syndrome in mice.

Using induced pluripotent stem cells (iPSCs) produced from the skin of a patient with a rare, genetic form of insulin-dependent diabetes called Wolfram syndrome, researchers transformed the human stem cells into insulin-producing cells and used CRISPR-Cas9 to correct a genetic defect that had caused the syndrome. They then implanted the cells into lab mice and cured the unrelenting diabetes in those models.
The findings, from researchers at Washington University School of Medicine in St. Louis, US, suggest this CRISPR-Cas9 technique may hold promise as a treatment for diabetes, particularly the forms caused by a single gene mutation and it also may be useful one day in some patients with the more common forms of diabetes, such as type 1 and type 2.
“This is the first time CRISPR has been used to fix a patient’s diabetes-causing genetic defect and successfully reverse diabetes,” said co-senior investigator Dr Jeffrey Millman, an assistant professor of medicine and of biomedical engineering at Washington University. “For this study, we used cells from a patient with Wolfram syndrome because, conceptually, we knew it would be easier to correct a defect caused by a single gene. But we see this as a stepping stone toward applying gene therapy to a broader population of patients with diabetes.”
Wolfram syndrome is caused by mutations to a single gene, providing the researchers an opportunity to determine whether combining stem cell technology with CRISPR to correct the genetic error also might correct the diabetes caused by the mutation.
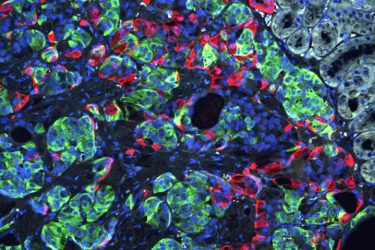

Researchers at Washington University School of Medicine in St. Louis have transformed stem cells into insulin-producing cells. They used the CRISPR gene-editing tool to correct a defect that caused a form of diabetes, and implanted the cells into mice to reverse diabetes in the animals. Shown is a microscopic image of insulin-secreting beta cells (insulin is green) that were made from stem cells produced from the skin of a patient with Wolfram syndrome [credit: Millman lab Washington University].
Millman and his colleagues had previously discovered how to convert human stem cells into pancreatic beta cells. When such cells encounter blood sugar, they secrete insulin. Recently, these researchers developed a new technique to more efficiently convert human stem cells into beta cells that are considerably better at controlling blood sugar.
In this study, they took the additional steps of deriving these cells from patients and using the CRISPR-Cas9 gene-editing tool on those cells to correct a mutation to the gene that causes Wolfram syndrome (WFS1). Then, the researchers compared the gene-edited cells to insulin-secreting beta cells from the same batch of stem cells that had not undergone editing with CRISPR.
In the test tube and in mice with a severe form of diabetes, the newly grown beta cells that were edited with CRISPR more efficiently secreted insulin in response to glucose. Diabetes disappeared in mice with the CRISPR-edited cells implanted beneath the skin and the animals’ blood sugar levels remained in normal range for the entire six months they were monitored. Animals receiving unedited beta cells remained diabetic. Although their newly implanted beta cells could produce insulin, it was not enough to reverse their diabetes.
“We basically were able to use these cells to cure the problem, making normal beta cells by correcting this mutation,” said co-senior investigator Dr Fumihiko Urano, the Samuel E. Schechter Professor of Medicine and a professor of pathology and immunology. “It’s a proof of concept demonstrating that correcting gene defects that cause or contribute to diabetes – in this case, in the Wolfram syndrome gene – we can make beta cells that more effectively control blood sugar. It’s also possible that by correcting the genetic defects in these cells, we may correct other problems Wolfram syndrome patients experience, such as visual impairment and neurodegeneration.”
“We’re excited about the fact that we were able to combine these two technologies – growing beta cells from induced pluripotent stem cells and using CRISPR to correct genetic defects,” Millman said. “In fact, we found that corrected beta cells were indistinguishable from beta cells made from the stem cells of healthy people without diabetes.”
Moving forward, the process of making beta cells from stem cells should get easier, the researchers said. For example, the scientists have developed less intrusive methods, making iPSCs from blood and they are working on developing stem cells from urine samples.
The study is published in Science Translational Medicine.
Related topics
CRISPR, Drug Targets, Genome Editing, Genomics, Induced Pluripotent Stem Cells (iPSCs), Personalised Medicine, Research & Development, Stem Cells
Related conditions
Diabetes, Wolfram syndrome
Related organisations
Washington University School of Medicine in St. Louis
Related people
Dr Fumihiko Urano, Dr Jeffrey Millman