Novel antiplatelet drug is effective at treating heart attacks in mice, say researchers
Posted: 16 July 2020 | Hannah Balfour (Drug Target Review) | No comments yet
The novel nanoparticle delivered Mp6 peptide was able to inhibit thrombosis and reduce inflammation without increasing the risk of bleeding in mice after heart attacks.
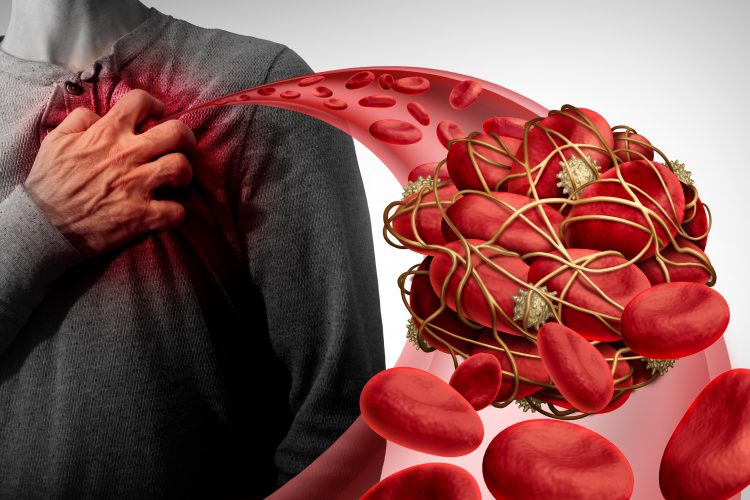
Scientists have developed a novel drug and nanoparticle delivery system to inhibit thrombosis (the formation of blood clots) without increasing the risk of bleeding. The team suggest the novel therapeutic could minimise the adverse effects of current antiplatelet medications, used as part of recovery after a heart attack or stroke.
Researchers at the University of Illinois at Chicago (UIC) demonstrated their drug was effective at treating heart attacks in animal models, without causing excessive bleeding.
Xiaoping Du, UIC professor of pharmacology and regenerative medicine at the UIC College of Medicine, who led the research published in Science Translational Medicine, said: “Unfortunately, current antiplatelet medications prevent the blood clotting that cause heart attack and stroke but also disrupt platelets’ ability to stop bleeding if a blood vessel is torn. In some cases, severe bleeding can be life-threatening. The magic of this new drug is it prevents clots but does not make people prone to bleeding, which other drugs have failed to do.”
In a previous study, Du and colleagues identified that the integrin signalling mechanism involved in blood clotting did not require platelets to adhere to a wound to prevent bleeding. They developed a synthetic peptide (mP6) targeting the integrin signalling pathway that was able to prevent blood clotting, without causing bleeding. However, it was hindered by inefficient and impractical intracellular delivery.
In this latest study, the team developed a nanoparticle to deliver the mP6 peptide to platelets, the new peptide-derived nanoparticle drug is called M3mP6 high-loading peptide nanoparticle (HLPN). They then tested it in mice as a potential heart attack treatment.
Du explained that a heart attack can cause heart failure and death in two different ways: firstly, through initial damage by the clot blocking blood flow and oxygen supply, which can be treated via angioplasty, stent insertion and antiplatelet drugs; and secondly, when fresh blood flowing into the damaged heart tissue following the reopening of the artery triggers inflammation, causing leaks and clots in small blood vessels and further damage to the heart.
“This is called reperfusion injury,” Du said. “We were hopeful that this new drug, which does not cause blood vessel leaks, would help limit reperfusion injury and reduce the chance of heart failure and death, and our hypothesis was proved correct – we saw very promising results from our study.”
In the study, among the group of mice that received the injected treatment, there was reduced damage to the heart, reduced thrombosis and reduced inflammation. There also was improved heart function and improved survival. The HLPNs loaded with engineered mP6 also demonstrated therapeutic effects in a model of ischemia-reperfusion injury.
“It is very exciting to see such promising results in the lab and we hope to one day test this in humans,” Du concluded.
Related topics
Drug Delivery, Drug Development, Drug Targets, In Vivo, Nanoparticles, Peptide Therapeutics, Therapeutics
Related conditions
Heart attack, inflammation, Ischemic stroke, thrombosis
Related organisations
University of Illinois at Chicago (UIC)
Related people
Xiaoping Du