Team redesigns COVID-19 Spike protein for more stable vaccines
Posted: 24 July 2020 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have modified the COVID-19 Spike protein for use in vaccines, which produces up to 10 times more protein than that of an earlier synthetic versions.

A team at The University of Texas (UT) at Austin, US, has successfully redesigned a key protein from SARS-CoV-2, the virus causing the COVID-19 pandemic. They say the modification could enable much faster and more stable production of vaccines to tackle the novel coronavirus worldwide.
Most COVID-19 vaccine candidates train the human immune system to recognise a key protein on the surface of the SARS-CoV-2 virus called the Spike (S) protein in order to fight infection. However, the UT researchers designed a new version of this protein that, when expressed in cells, produces up to 10 times more protein than that of an earlier synthetic S protein already in use in multiple COVID-19 vaccine candidates.
“Depending on the type of vaccine, this improved version of the protein could reduce the size of each dose or speed up vaccine production,” said Jason McLellan, an associate professor in the Department of Molecular Biosciences and senior author of the paper. “Either way, it could mean more patients have access to vaccines faster.”
Biomarkers aren’t just supporting drug discovery – they’re driving it
FREE market report
From smarter trials to faster insights, this report unpacks the science, strategy and real-world impact behind the next generation of precision therapies.
What you’ll unlock:
- How biomarkers are guiding dose selection and early efficacy decisions in complex trials
- Why multi-omics, liquid biopsy and digital tools are redefining the discovery process
- What makes lab data regulatory-ready and why alignment matters from day one
Explore how biomarkers are shaping early drug development
Access the full report – it’s free!
Dubbed HexaPro, the new protein is also more stable than an earlier version the team made of the S protein, which they say should make it easier to store and transport. It also keeps its shape even under heat stress, during storage at room temperature and through multiple freeze-thaws. According to the researchers, such qualities are desirable in a robust vaccine.
Drawing on their experience creating stabilised proteins as vaccines against MERS-CoV, the coronavirus that causes Middle East respiratory syndrome, and other viruses, the researchers identified 100 different modifications to the S protein that they believed might lead to a more stable, more highly expressed version. Next they created 100 different versions of the protein by inserting the genetic blueprints for each version into a different culture of human cells. Of those 100 versions of the S protein, 26 were more stable or had higher expression.
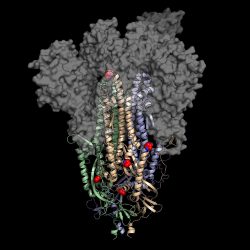

This is a 3D atomic scale map, or molecular structure, of Hexapro, a modified version of the SARS-CoV-2 spike protein, described in a paper in the journal Science on July 23, 2020. Six key modifications, called prolines, are indicated as red and blue spheres and help lock the protein into the prefusion conformation, the shape it takes before infection [credit: University of Texas at Austin].
The researchers then took four of those beneficial modifications, plus two from their original stabilised S protein and combined them to create HexaPro. When they inserted the genetic blueprints for this version of the S protein into a human cell culture, the cells produced 10 times as much protein than that of their original protein.
The team add that HexaPro could also be used in COVID-19 antibody tests where it would act as a probe to identify the presence of antibodies in a patient’s blood, indicating whether a person has previously been infected with the virus.
For nucleic acid-based vaccines that use the patient’s own cells to create the viral proteins that trigger an immune response, such as mRNA-1273, the team say this improved S protein might enable next-generation versions that require a much smaller dose to elicit the same immune response from a patient. For subunit vaccines that contain a version of the actual viral protein as an antigen, such as NVX-CoV2373, many more vaccine doses could be produced in the same time frame.
The new findings are described in the journal Science.
Related topics
Drug Development, Drug Targets, Immunology, Protein, Proteomics, Research & Development, Structural Biology, Vaccine
Related conditions
Coronavirus, Covid-19, Middle East Respiratory Syndrome (MERS)
Related organisations
The University of Texas at Austin
Related people
Jason McLellan








