Stem cell therapy repairs damage from dementia in mouse brains
Posted: 22 April 2021 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have developed a stem cell therapy that could treat the damage caused by dementia by launching a repair response.


A one-time injection of an experimental stem cell therapy was shown to repair brain damage and improve memory function in mice with conditions that replicate human strokes and dementia, researchers have revealed. The study was conducted at the University of California, Los Angeles (UCLA), US.
According to the researchers, dementia can arise from multiple conditions and it is characterised by problems with memory, attention, communication and physical co-ordination. The two most common causes of dementia are Alzheimer’s disease and white matter strokes – small strokes that accumulate in the connecting areas of the brain.
“It is a vicious cycle; the two leading causes of dementia are almost always seen together and each one accelerates the other,” said Dr S Thomas Carmichael, senior author of the study.
In the new study, the team identified a cell therapy that can stop the progressive damage caused by the disease and stimulate the brain’s own repair processes.
The cells used in the therapy are a specialised type of glial cells, which are cells that surround and support neurons in the central nervous system (CNS). The team evaluated the effects of their glial cell therapy by injecting it into the brains of mice with brain damage similar to that seen in humans in the early to middle stages of dementia.
“Upon injection, our cell therapy travelled to damaged areas of the brain and secreted chemicals called growth factors that stimulated the brain’s stem cells to launch a repair response,” said Dr Irene Llorente, the paper’s first author.
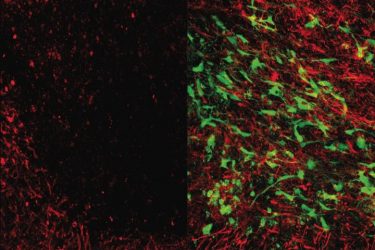

Microscope images showing brain tissue that has been damaged by white matter stroke (left) and then repaired by the new glial cell therapy (right). Myelin (seen in red), is a substance that protects the connections between neurons and is lost due to white matter stroke. As seen at right, the glial cell therapy (green) restores lost myelin and improves connections in the brain [credit: UCLA Broad Stem Cell Research Center/Science Translational Medicine].
Activating that repair process not only limited the progression of damage, but also enhanced the formation of new neural connections and increased the production of myelin – a fatty substance that covers and protects the connections.
The researchers found that even if they eliminated the injected cells a few months after they had been transplanted, the mice’s recovery was unaffected. That is because the therapy primarily serves as a wake-up call to stimulate the brain’s own repair processes.
“Understanding the role that glia play in repairing white matter damage is a critically important area of research that needs to be explored,” said Francesca Bosetti, a programme director at the US National Institutes of Health’s (NIH) National Institute of Neurological Disorders and Stroke, which supported the study. “These preliminary results suggest that glial cell-based therapies may one day help combat the white matter damage that many stroke and vascular dementia patients suffer every year.”
The team used a method for quickly producing large numbers of glial cells by treating human induced pluripotent stem cells (iPSCs) with a drug called deferoxamine. iPSCs are derived from skin or blood cells that have been reprogrammed back to an embryonic stem cell-like state from which scientists can create an unlimited supply of any cell type.
In the future, if the therapy is shown to be safe and effective through clinical trials in humans, the researchers envision it becoming an “off-the-shelf” product, meaning that the cells would be mass manufactured, frozen and shipped to hospitals, where they could be used as a one-time therapy for people with early signs of white matter stroke.
“The damage from white matter strokes is progressive, so you do not have months to spend producing a treatment for each patient,” said Carmichael. “If you can have a treatment that is already in the freezer ready to go during the window of time when it could be most effective, that is a much better option.”
The team is now conducting the additional studies necessary to apply to the US Food and Drug Administration (FDA) for permission to test the therapy in a clinical trial in humans.
The results are published in Science Translational Medicine.
Related topics
In Vivo, Induced Pluripotent Stem Cells (iPSCs), Neurosciences, Regenerative Medicine, Stem Cells, Translational Science
Related conditions
Alzheimer’s disease, Dementia, Stroke
Related organisations
University of California Los Angeles (UCLA), US National Institutes of Health (NIH)
Related people
Dr Irene Llorente, Dr S Thomas Carmichael, Francesca Bosetti