PP2A enzyme could be major driver of preeclampsia
Posted: 10 September 2021 | Anna Begley (Drug Target Review) | No comments yet
A new study found that protein phosphatase 2 (PP2A) found to be a major driver of preeclampsia, potentially leading to treatments.

A new study by University of Texas (UT) Southwestern, US, indicates that the enzyme protein phosphatase 2 (PP2A) appears to be a major driver of preeclampsia, a dangerous pregnancy complication characterised by the development of high blood pressure and excess protein in urine. The findings could lead to new treatments for preeclampsia other than premature delivery, which is often the only option.
“Preeclampsia is a regrettably common cause of premature birth, which can be life-threatening for babies and lead to lifelong consequences,” explained study leader Professor Philip Shaul. “Through identifying PP2A’s role in this condition, we may be able to develop treatments for preeclampsia that are far better for both mothers and babies.”
Although the causes of preeclampsia are not well understood, researchers have linked the condition to a variety of risk factors. One is an autoimmune disease known as antiphospholipid syndrome (APS), in which antibodies react to proteins on the surface of some cells. Although APS is relatively rare, affecting only about five in every 100,000 people, studies have identified APS antibodies in about 29 percent of pregnant women with preeclampsia.
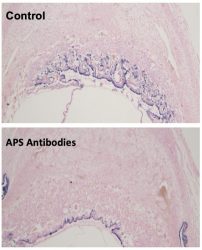

Images shows how APS antibodies inhibit trophoblast migration in the mouse placenta [credit: UT Southwestern Medical Centre].
To better understand how APS leads to preeclampsia, the team created an animal model by injecting pregnant mice with APS antibodies. These animals developed high blood pressure and a rise in urine protein, which are characteristics of preeclampsia. In contrast, the APS antibodies did not affect blood pressure in non-pregnant mice.
Based on previous work, the researchers knew that a protein called ApoER2 may be related to the harmful actions of APS antibodies on placental cells called trophoblasts which do not successfully make that connection in preeclampsia. In mice, the APS antibodies prevented trophoblast migration and growth of the foetus was restricted. When the researchers genetically engineered mice without ApoER2 in trophoblasts, the foetuses developed normally despite APS antibody treatment and the mothers were protected from developing preeclampsia.
The researchers also found that in the presence of the APS antibodies, ApoER2 triggers the activity of PP2A, an enzyme that regulates protein functions throughout the body. Further experiments showed that in the pregnant mice with APS antibodies, heightened activity in PP2A increased trophoblast production of proteins known to be involved in preeclampsia. When the researchers gave the pregnant mice a drug that inhibits PP2A, they were protected from preeclampsia and the treatment had no apparent harmful effects on the mice or their gestating babies.
The scientists then examined placentas from women with APS, finding that they had increased activity of PP2A. Moreover, they discovered that compared with placentas from normal pregnancies, those from preeclamptic patients without APS also had increased PP2A activity, suggesting that this mechanism could be operating in a variety of forms of preeclampsia. The team hope that with further research treatments targeting PP2A or its related machinery in the trophoblast may eventually be viable treatments for preeclampsia in pregnant women.
The findings were published in Circulation Research.
Related topics
Antibodies, Drug Targets, Enzymes, In Vivo, Molecular Biology, Molecular Targets, Protein, Protein Expression, Small molecule
Related conditions
Preeclampsia
Related organisations
University of Texas Southwestern (UT Southwestern)
Related people
Professor Philip Shaul