Celltrion selects 14 lead monoclonal antibodies for COVID-19 treatment
Posted: 16 April 2020 | Victoria Rees (Drug Target Review) | No comments yet
After screening 300 antibodies, Celltrion has identified the 14 most powerful that could potentially combat the COVID-19 coronavirus.

Celltrion has announced it has completed its hit-to-lead stage of drug discovery to identify 14 powerful neutralising monoclonal antibodies (mAbs) against SARS-CoV-2, the virus that causes COVID-19.
These antibodies can recognise multiple epitopes, thus increasing the probability of neutralisation against viral mutations”
Through a partnership with the Korea Centers for Disease Control and Prevention (KCDC), Celltrion initially identified and secured 300 different types of antibodies that bind to the SARS-CoV-2 antigen. These were then screened based on their ability to bind to the virus Spike (S) protein. Celltrion was then able to capture a total of 38 potent neutralising antibodies, of which, 14 were identified as most potent against SARS-CoV-2.
Following the selection of antibody candidates which demonstrate high potency in neutralising SARS-CoV-2, Celltrion will now begin cell-line development. Once this is completed, the company aims to roll out mass production of the therapeutic antibody and together with the KCDC, Celltrion will conduct efficacy and toxicity testing in mice and non-human primates.
Ki-Sung Kwon, Head of R&D Unit at Celltrion said: “We are bringing our full resources and expertise to overcome this global health crisis and are glad to have identified these antibodies sooner than previously expected. These antibodies can recognise multiple epitopes, thus increasing the probability of neutralisation against viral mutations. Given the expedited development process of our antiviral antibody treatment, we anticipate moving to first-in-human clinical trials in July. We are also on track with the development of a ‘super antibody’ or ‘an antibody cocktail’ and the launch of a rapid diagnostic kit in the summer of this year.”
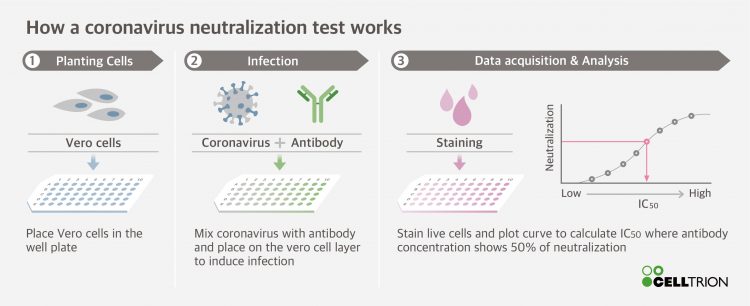

The antibody neutralisation test process [credit: Celltrion].
Related topics
Antibodies, Antibody Discovery, Hit-to-Lead, Research & Development, Screening, Targets
Related conditions
Coronavirus, Covid-19
Related organisations
Celltrion, Korea Centers for Disease Control and Prevention (KCDC)
Related people
Ki-Sung Kwon