Scientists identify protein that prevents HSV-1 brain damage
Posted: 20 October 2021 | Anna Begley (Drug Target Review) | No comments yet
Mammalian target of rapamycin complex 2 (mTORC2) was found to prevent brain damage in mice infected with herpes simplex virus 1 (HSV-1).
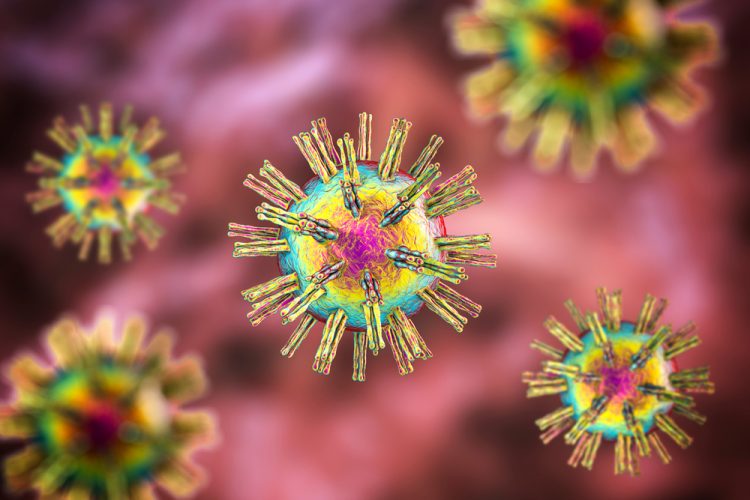

A team at the University of Illinois Chicago, US, have that the protein mammalian target of rapamycin complex 2 (mTORC2) limits herpes simplex virus 1 (HSV-1) infection through rapid activation of antiviral immunity and protects the host by preventing encephalitis and possible death. The team hope that their findings will encourage other researchers to investigate mTORC2 for future treatments.
Ocular herpes infection is a leading cause of infectious blindness and can lead to lethal brain infections. After a primary infection, HSV-1 hides in neuronal tissues and reactivates under immune suppressive conditions that may cause encephalitis, leading to permanent brain damage, memory loss or even death. Herpes is a common infection and is rarely lethal. To understand how antiviral defence mechanisms could work, the researchers studied mTORC2, explained Professor Deepak Shukla who led the study, published recently in Nature Communications.
“Animals with a lack of functional mTORC2 were found to show significant loss of immune activation and more spread of virus to neuronal tissues,” Shukla commented. “Functional mTORC2 limits the virus spread from ocular to neuronal tissues by supporting the production of antiviral cytokines and driving immune cells to quickly identify HSV-1 infected cells. MTORC2 also protects the ocular and neuronal cells from virus-induced cell death offering protection to neuronal tissue.”
In order to combat lifelong HSV-1 infection, it is essential to uncover the role of key cellular proteins and modulate their function to stop the virus from replicating and causing damage, added Rahul Suryawanshi, the study’s lead author. During herpes infection, mTORC2 activates cell survival machinery in ocular and neuronal tissues. This restricts a programmed cell death mechanism, which limits virus spread but, at the same time, is detrimental to neuronal cells.
NEWS: Statistical model defines how ketamine anaesthesia affects the brain – READ HERE
“The animal experiments highlight the importance of using neuronal cell death inhibitors during viral encephalitis that may mimic and/or improve mTORC2 functions and prevent brain injuries,” said co-author Chandrashekhar Patil.
Shukla stated that the study explains that the pro-survival functions of mTORC2 may not be limited to viral infections and are likely to be applicable to many other diseases. “Our study will motivate other researchers to investigate mTORC2 in neurodegenerative disorders and diseases as well,” he concluded.
Related topics
Drug Leads, Immunology, In Vivo, Neurons, Neuroprotection, Neurosciences, Protein, Small Molecules, Therapeutics
Related conditions
Herpes simplex virus (HSV-1)
Related organisations
University of Illinois at Chicago (UIC)
Related people
Chandrashekhar Patil, Deepak Shukla, Rahul Suryawanshi