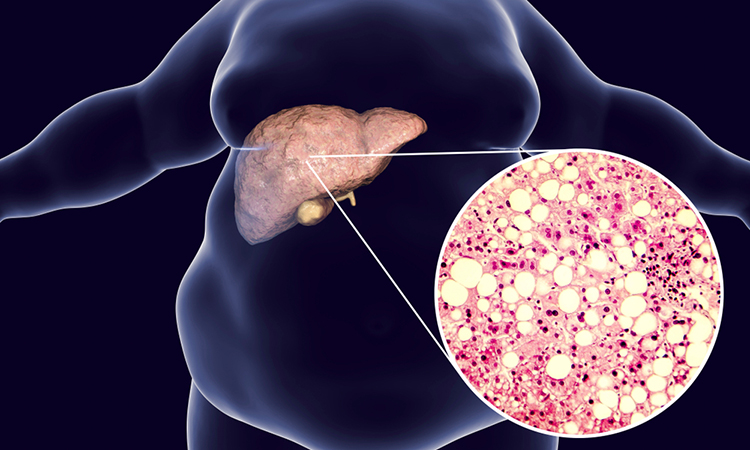
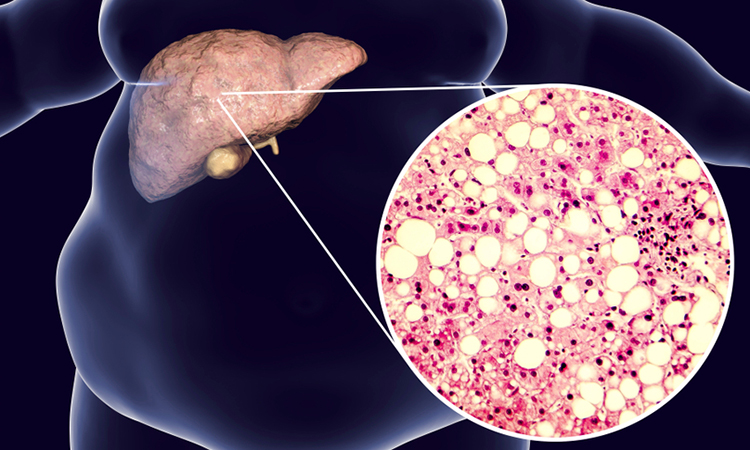
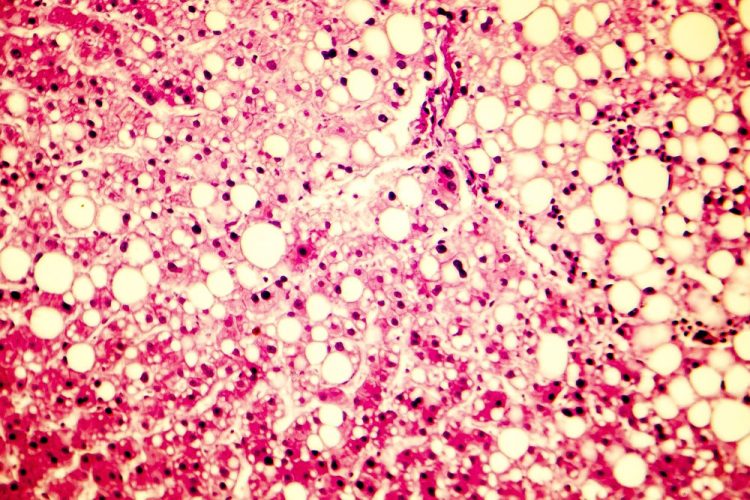
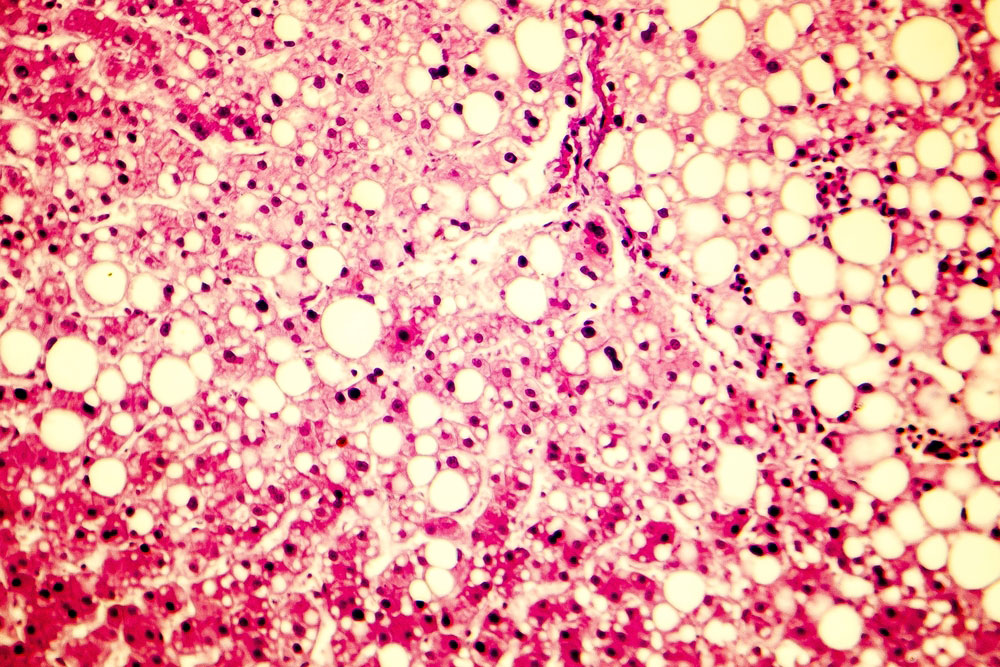
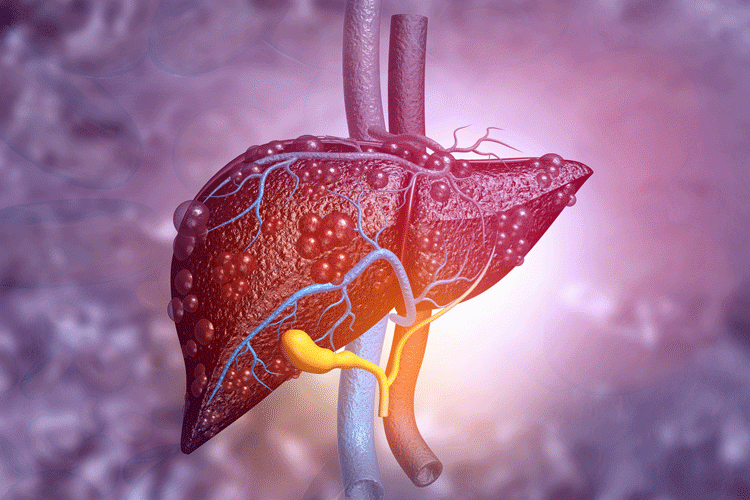
Researchers develop organoid models to study non-alcoholic fatty liver disease
The team used these models to show drug responses and established a CRISPR-screening platform to identify potential therapeutic targets for non-alcoholic fatty liver disease.