Experimental HIV vaccine induces bnAb production in models
Posted: 21 November 2019 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have developed a vaccine for HIV that has shown success in rabbits, by neutralising a wide range of HIV strains using bnAbs.
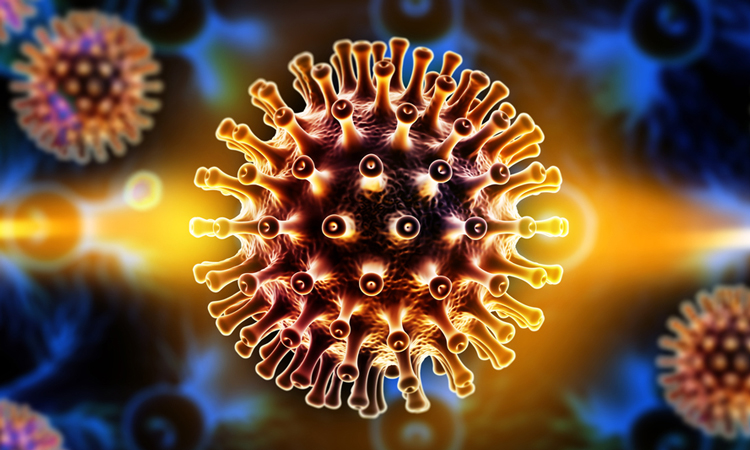

An experimental HIV vaccine has shown success in pre-clinical trials by inducing antibody production which neutralises a wide range of HIV strains.
The study was conducted at Scripps Research, US, in collaboration with non-profit vaccine research organisation IAVI.
The researchers demonstrated that the vaccine was able to elicit broadly neutralising antibodies (bnAbs) in rabbits, which targeted at least two critical sites on the virus. The bnAbs are able to work against HIV as they bind to sites that vary little from strain to strain. It is considered by most researchers that bnAbs need to neutralise multiple sites on HIV to provide robust protection.
The team are continuing to test and improve their vaccine strategy in small animal models”
Although patients with HIV can produce bnAbs as part of their antibody response, this can often be infrequent and after the infection has long been established. Therefore, vaccine designs for HIV have revolved around ways to stimulate the immune system into making bnAbs.
In the current study, the researchers designed the vaccine as a virus-mimicking protein based on the ‘Env’ protein in HIV. Multiple copies of bush-like Env proteins are usually spread out on the surface of each spherical HIV particle.
Each Env protein contains a molecular mechanism that allows it to bind to a receptor on immune cells known as CD4 and use that receptor as a portal to break into the cell. The researchers engineered a version of Env that models its essential structures while being stable enough to use as a vaccine. To present it in a way that would resemble a real HIV virus particle, they created virus-sized liposomes which were studded densely with the engineered Env proteins.
On a natural HIV Env protein, groups of sugar-related molecules called glycans help to shield the CD4 binding site from immune attack. As an initial immunisation strategy, the researchers used versions of Env in which this glycan shield around the CD4 binding site had been partly removed.
Subsequent booster immunisations over 48 weeks used Env proteins with restored glycans to select antibodies that target the CD4 binding site but can also penetrate this shield. The Env proteins in the booster shots were mixes based on different strains of HIV, to generally promote antibody responses against Env structures that do not vary among these strains.
The team inoculated 12 rabbits after designing their vaccine strategy and compared the results with a control group that received only a single, glycan-shielded version of Env. They found that their strategy had a much better response, with five of the rabbits developing antibodies that could neutralise multiple HIV isolates.
The researchers analysed the antibodies of the rabbit that had responded most strongly and identified two distinct types of bnAb. One, which they called E70, blocks the CD4 binding site, partly by binding to one of the shielding glycans. The other, 1C2, hits a different but vulnerable spot on Env, at the interface between two key segments of the complex protein. The binding of 1C2 destabilises Env so that it can no longer mediate HIV’s entry into host cells. 1C2 was found to have an unusual breadth of neutralisation, blocking 87 percent of a panel of 208 distinct HIV isolates.
The team are continuing to test and improve their vaccine strategy in small animal models and hope eventually to test it in monkeys and then humans.
“It’s an initial proof-of-principle but an important one and we’re now working to optimise this vaccine design,” says the study’s senior author Dr Richard Wyatt, a professor in the Department of Immunology and Microbiology at Scripps Research.
The results were published in Immunity.
Related topics
Antibodies, Drug Development, Drug Targets, Research & Development, Targets, Vaccine
Related conditions
HIV
Related organisations
IAVI, Scripps Research
Related people
Dr Richard Wyatt