Novel antibody-drug candidate to treat pancreatic cancer developed
Posted: 10 November 2020 | Victoria Rees (Drug Target Review) | No comments yet
Scientists have developed a new antibody-drug conjugate (ADC), made from ICAM1, an antibody that targets pancreatic cancer and the cytotoxic drug DM1 (mertansine).

A pre-clinical study has used a highly selective, potent, engineered antibody-drug combination to induce marked and lasting tumour regression in a mouse model of pancreatic cancer. The study was conducted at Boston Children’s Hospital, US.
According to the scientists, pancreatic cancer has proven very difficult to treat with drugs. Not only do the tumours have a limited blood supply, making it hard to deliver drugs, they are also surrounded by stroma (a dense connective tissue) and tumour cells are protected by an extracellular matrix (a meshwork of proteins and carbohydrates).
“It can be difficult to get drugs into these tumours,” said Dr Marsha Moses, whose lab the research was conducted in. “We developed a novel chemo-immunotherapy agent that selectively recognises and penetrates pancreatic tumours better than other therapeutics.”
Led by Dr Jing Huang, the Moses lab developed an antibody-drug conjugate (ADC), consisting of two parts: an antibody that selectively homes to a molecule on the surface of pancreatic cancer cells, known as ICAM1 and a drug toxic to cancer cells. The team chose ICAM1 as a target for the ADC antibodies after screening the tumour surface for dozens of different proteins.
The team performed similarly unbiased screening to select the best drug to include in the ADC from a pool of drugs already used clinically. They tested four candidate ADCs in two human pancreatic cancer cell lines as well as in normal pancreatic cells. ADCs combining ICAM1 antibodies with the cytotoxic drug DM1 (mertansine), used clinically in HER2-positive breast cancer, were the most effective in killing tumour cells, working better than other ADC drugs. The DM1-ICAM1 antibody combination did not harm non-cancerous pancreatic cells which do not produce ICAM-1.
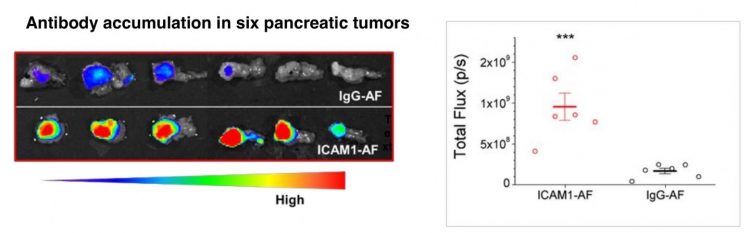

At left, the red staining shows high levels of ICAM1 antibody accumulation in human pancreatic tumours with adjacent normal pancreas tissues. The blue staining shows low accumulation of non-targeting IgG antibodies in the tumours. At right, quantification of antibody accumulation [credit: Dr Jing Huang, Boston Children’s Hospital].
The team next randomised mice with pancreatic tumours to receive one of four treatments, given as systemic injections: the DM1-ICAM1 antibody conjugate, DM1 bound to a non-targeting antibody (IgG), gemcitabine (a first-line chemotherapy drug used in pancreatic cancer) or a placebo.
Compared with the other groups, the researcher found that the mice receiving the DM1-ICAM1 antibody conjugate had a significant reduction in tumour size that persisted during the 14-week study, even after just two doses. The treatment also effectively inhibited metastasis to normal organs including lung, liver and spleen. There was no observed toxicity, assessed by weighing the animals and through pathology analysis of their organs.
Finally, Huang developed an MRI-based molecular tumour imaging technique to complement ICAM1 ADC therapy, confirming the presence of ICAM1 on the tumour without the need for an invasive biopsy. The team say this could potentially help to predict the treatment’s effect and monitor changes over time.
“The size of the ADC is similar to the size of a single antibody: less than 10 nanometers,” said Dr Peng Guo, co-corresponding author. “Because of this ultra-small diameter, it can penetrate the stroma and reach pancreatic tumour cells better than other novel treatments such as T-cell immunotherapy or nanomedicines.”
“The precision of our approach comes from both the specific targeting and the ability to monitor that targeting with MRI,” Huang said.
The study was published in Advanced Science.
Related topics
Antibodies, Antibody Discovery, Drug Development, Drug Leads, Drug Targets, Research & Development, Screening, Targets, Therapeutics
Related conditions
HER2-driven breast cancer, Pancreatic cancer
Related organisations
Boston Children's Hospital
Related people
Dr Marsha Moses, Dr Peng Guo